The Periodic Law (Periodic Table)
430 likes | 557 Vues
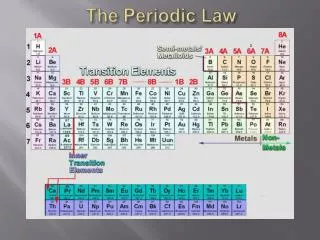
The Periodic Law (Periodic Table). CHAPTER FIVE. Brief History. Original periodic table was constructed by Dmitri Mendeleev . This periodic table consisted of about 60 elements and was arranged in order of increasing atomic mass. Modern Periodic Table.

The Periodic Law (Periodic Table)
E N D
Presentation Transcript
The Periodic Law (Periodic Table) CHAPTER FIVE
Brief History Original periodic table was constructed by Dmitri Mendeleev. This periodic table consisted of about 60 elements and was arranged in order of increasing atomic mass
Modern Periodic Table • Our periodic table is based on the work of Henry Mosely, who arranged the elements in order of increasing atomic number
Periodic Law • Mosely’s and Mendeleev’s periodic table follows periodic law: • This means that if elements are arranged in increasing atomic #, they will fall into groups that have similar properties.
Modern Periodic Table • 18 vertical group/families • 7 horizontal periods • Divided into two main parts, metals and nonmentals.
Left Side of the periodic table • Metals • Shiny • Malleable • Ductile • Conducts electricity • Conducts Heat • Mostly solids
Group 1 Elements • Alkali Metals • Most reactive of the metals • Mostly Soft Solids
Group 2 Elements • Alkaline Earth Metals • Still very reactive • More dense than the group 1 metals
Groups three through twelve • Transitional Metals • Elements “transition” from Metals to non-metals as you move across the table • Most Transition metals have metallic properties
The “Others” • Groups 13-16 • Other Metals (Have metallic properties) • Al, Ga, In, Sn, Bi, Pb, Tl • Metalloids-Boron, Silicon, Germanium, Arsenic, Antimony, Tellurium • Properties of both metals and non-mentals • Semiconductors-used in electronics • Other Non-Metals (Have non-metal properties) • C, N, O, P, S, Se, H
Right side of periodic table • Non-Metals • Do not conduct electricity or heat well • Some brittle solids • Some gasses
Group 17 • Halogens • Non-metal • most reactive of nonmetals.
Group 18 • Noble gases • Nonmetals • Least reactive of non-metals
Check for understanding Based on the group/family each element below are in, name 2 properties of : 9. K 10. P 11. Cl 12. Ga Which group/family is each element in? • Al • Ar • K • Pd • Br • Ca • Zn • Si
Periodic Trends The tendencies of certain elemental characteristics to increase or decrease as one progresses along a row or column of the periodic table of elements.
Atomic Radii • Atomic Radii-The distance from the center of the nucleus to the edge of the atom’s electron cloud. Atomic Radii
Atomic Radii Trending Activity • Grapher • Straw Cutter • Recorder • We will use our current knowledge of the atom to try and predict the trend for atomic radii. • We will then test our hypothesis by actually graphing real data and then making a 3-D model of the trend.
Atomic Radii Continued • Trend in each period • Why do you think the radius decreases to the right if there are more protons to the right? Shouldn’t that make the radius increase? • As there are more protons they pull more strongly on the electrons that are surrounding them. • Each period is within the same energy level.
Atomic Radii Continued • Trend in each group • Why do you think the radius increases as you go down a group if more protons means they pull harder on the electrons? • As you go down a group you are adding energy levels which makes the radius increase.
Atomic Radii Continued • Periodic Table Trend: • Increases to the left • Increases going down Increasing Size
Atomic Radii Practice In each pair which of the following has the largest Atomic Radii? • K or Ni • Ne or Xe • Rb or In Arrange the following in order of increasing atomic radii? 4. Al, K, Ne, H • Answer: H, Ne, Al, K, 5. Will a larger atomic radii make an atom more or less likely to give away an electron?
Quiz • Place the following in order using decreasing Atomic Radii : Au, Ba, O, Cl & Pb • Cl or I • Which of the above pair has the largest atomic radii? • Why does the table trend that way? • Who created the 1st periodic table? • What was the 1st periodic table arranged by? • The vertical columns are called what? • Elements within the same column will have _________ properties. • What is the name of each of the following groups? • 18 • 2 • 1 • Which group name does Ge fall under? • Name 4 popertiesA
Ionization Energy (IE) • 1st Ionization Energy-Energy required to remove one electron from an un-bonded atom. (individual atoms) • Basically: How hard is it to get an electron away from an atom? No, Mine! Mine! e-
Ions • If enough energy is input into an atom, an electron will be removed. This forms an Ion. • ION-an atom that has a positive or negative charge.
Ionization Energy (IE) • A lost electron will create a Positive Ion • Positive Ion is a Cation + + + + + + + Pawsitive : )
Ionization Energy (IE) • A gained electron will create a negative ion • Negative ions are called Anions
Define Valence Electrons: Define Lewis Dot Noation: In the above configurations underline the highest energy level represented (not the orbitals, just the energy levels) Circle the electrons in each of those highest energy levels Write the number of valence electrons in the correct column Draw the Lewis Dot Notation in the correct column Draw the Borh Model for each element in the correct column Define Shielding Effect:
Valence Electrons and Ionization Energy Make the same chart for each of the elements in the following Groups as well • Group 2 • Be • Ca • Ba • Group 13 • B • Al • Ga • Group 14 • C • Si • Group 15 • N • P • Group 16 • O • S • Group 17 • F • Cl • Group 18 • He • Ne • Ar
Ionization Energy (IE) Continued • Trend across a period? • Decreases to the left • Trend in a group/family? • Decreases going down • Which Group would have the lowest first Ionization energy? (who is it easiest to take an electron from?) • Group 1 • Why? • They want to get rid of that 1 valence electron • Which group would have the highest first ionization energy? • Group 18 • Why? • They do not want to lose one of their 8 valence electrons ( they are happy the way they are)
Ionization Energy • Periodic Table Trend: • Decreases to the left • Decreases going down Decreasing Decreasing
Ionization energy practice • Which element within the following pairs has the highest ionization energy? • K or Be • B or Ga • O or Ca • Ar or Sb • Arrange the following in decreasing Ionization energy (meaning from Highest to lowest) • Li, O, C, K, Ne, F
Electron Affinity • Electron Affinity-The energy change that occurs when an individual atom gains an electron. • The more an atom “loves” getting another electron the more the energy it will release. . . _ . . + Electron Atom
Electron Affinity • Which group is “happiest” to take on an electron? • Group 17 Why? • They are 1 electron away from a full outer shell (Valance Shell)
Electron Affinity • Which groups care nothing for additional electrons? • Group 1 and Group 18? Why? • Group 18 is totally happy with their 8 electrons • Group 1 is closer to being happy by giving away it’s 1 valance electron.
Electron Affinity • Periodic Table Trend: • Decreases to the left • Decreases going down Decreasing Decreasing
Electron Affinity • Which of the following has the greatest electron affinity? • Al or Br • O or C • Xe or F • Arrange by increasing Electron Affinity • Cl, K, Fr, I • Answer:Fr, K, I, Cl
Electronegativity • Electronegativity-Measurement of how strongly one atom attracts the electrons of another atom in a compound. • Developed by Linus Pauling • Which element do you think would be the most electronegative? • Fluorine-4.0. All other values are calculated in relation to Fluorine’s value. • Scientest just picked the number 4. It wasn’t really based on anything.
Electronegativity & (FON) • Oxygen, Nitrogen and then the other halogens are the most Electronegative after fluorine. • F, O, N – Top 3 highest electronegativity
Electronegativity • Periodic Table Trend: • Decreases to the left • Decreases going down Decreasing Decreasing
Electronegativity Practice • Which of each pair has the greatest EN? a. Ca or Ga b. Br or As c. Li or O d. Ba or Sr e. Cl or S f. O or S • Rank the following elements by increasing EN: sulfur, oxygen, neon, aluminum. • Answer: Ne,Al,S,O
Review 1. The elements characterized as nonmetals are located in the periodic table at the (A) left; (B) bottom; (C) center; (D) top right 2. Which is the atomic number of an alkali metal? (A) 10; (B) 11; (C) 12; (D) 13. 3. Which element is a halogen? (A) iron; (B) nitrogen; (C) iodine; (D) neon 4. Which Group 17 element has the least tendency to gain electrons? (A) fluorine; (B) iodine; (C) bromine; (D) chlorine. 5. The alkaline earth element having the largest atomic radius is found in Period (A) 1; (B) 2; (C) 6; (D) 7. 6. Which of the following atoms will lose an electron the easiest? (A) potassium; (B) calcium; (C) rubidium; (D) strontium