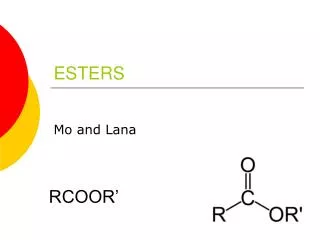
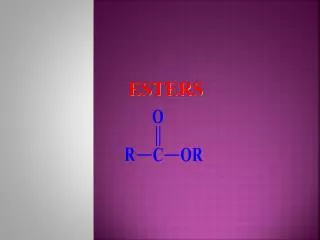
The Esters
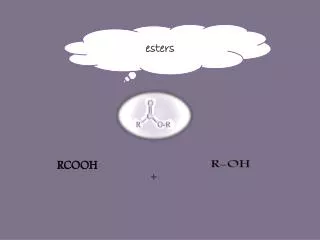
The Esters. Fischer Esterification. This is not necessarily the best method of preparing esters, but it does serve to illustrate that esters are essentially the products of a reaction between a carboxylic acid and an alcohol. Lactones.


The Esters
E N D
Presentation Transcript
The Esters WWU-Chemistry
Fischer Esterification This is not necessarily the best method of preparing esters, but it does serve to illustrate that esters are essentially the products of a reaction between a carboxylic acid and an alcohol. WWU-Chemistry
Lactones Lactones are cyclic esters formed by the intramolecularinteraction of an alcohol functional group with a carboxylic acid functional group: a lactone WWU-Chemistry
The Triacylglycerols • A very important class of esters, from a biological point of view, is in the triacylglycerols -- the common simple fats. • “Triacylglycerol” is a relatively modern term; you may also hear of these substances as “simple lipids” or as “triglycerides.” • The triacylglycerols are esters of long-chain carboxylic acids combined with glycerol. WWU-Chemistry
The Triacylglycerols The nature of the R groups can vary. WWU-Chemistry
Glycerol Since glycerol has three -OH groups, it can react with three molecules of carboxylic acid to form a triester. WWU-Chemistry
The Fatty Acids • The carboxylic acid components of triacylglycerols are known as fatty acids. • Fatty acids are simple, long-chain carboxylic acids -- their name comes from the simple fact that they occur in fats! • Naturally-occurring fatty acids always have: • an unbranched carbon chain • an even number of carbon atoms WWU-Chemistry
The Fatty Acids WWU-Chemistry
Saturated versus Unsaturated Fatty Acids • When the carbon chain (excluding the C=O bond) contains single bonds only, the fatty acid is known as a saturated fatty acid, and the triacylglycerol thus becomes a saturated fat. • When the carbon chain contains one or more C=C double bonds, the fatty acid becomes an unsaturated fatty acid, and the triacylglycerol thus becomes an unsaturated fat. WWU-Chemistry
Unsaturated Fatty Acids • Naturally-occurring unsaturated fatty acids have the following characteristics: • The first double bond appears at carbon #9 • If there are two or more double bonds, they are separated by a -CH2- group -- the double bonds are never conjugated. • Double bonds always have the cis geometry. The so-called trans fatty acids are not naturally-occurring. WWU-Chemistry
Hydrogenation of Fats WWU-Chemistry
Hydrogenation of Fats • The hydrogenation of an unsaturated fat converts it into a saturated fat. • This is an important process in the food industry, where unsaturated (liquid) fats are converted into saturated (solid) fats. • Often, in an effort to retain some of the nutritional benefits of an unsaturated fat, food manufacturers will only partially hydrogenate a liquid fat -- just sufficiently for the material to turn semi-solid. This would be a partially hydrogenated fat. WWU-Chemistry
Isomerization of Fatty Acids WWU-Chemistry
Olestra (“Olean”) WWU-Chemistry
Molecular Model of Triolein WWU-Chemistry
Molecular Model of Olestra WWU-Chemistry
Fischer Esterification This is the most direct way of preparing esters. WWU-Chemistry
Fischer Esterification • This is a reversible reaction; the equilibrium constant is only slightly greater than unity (between 1 and 10). • To be successful, this reaction requires that one be able to shift the equilibrium to the right • This is accomplished by: • removing water by azeotropic distillation • using an excess of whichever reagent is inexpensive. WWU-Chemistry
Fischer Esterification 84% yield WWU-Chemistry
Preparation of Esters(Best Method) WWU-Chemistry
Also Good: You would use this alternative when the acid anhydride is cheaper than the corresponding acid chloride. There are relatively few commercially-available acid anhydrides, however. WWU-Chemistry
Methyl Ester Formation • Carboxylic acids can be converted to methyl esters by the action of diazomethane.CH2N2 WWU-Chemistry
Methyl Ester Formation Notice that the CH2 of diazomethane appears to insert itself between the O and the H of the O-H bond! WWU-Chemistry
Diazomethane • Diazomethane is a highly toxic and dangerously explosive gas. • In spite of its hazardous nature, it can be handled relatively safely in dilute ether solutions (and in small quantities) WWU-Chemistry
Diazomethane(continued) • In dilute solution it is a very useful reagent for the preparation of methyl esters, particularly in cases where only small quantities of carboxylic acid are available. • This reaction proceeds in nearly quantitative (100%) yields! WWU-Chemistry
Structure of Diazomethane WWU-Chemistry
Reactivity of Diazomethane • The high reactivity of diazomethane arises from the fact that it possesses an exceedingly reactive leaving group, the nitrogen molecule (which is exceptionally stable). • As the following mechanism shows, a nucleophilic substitution reaction on the protonated diazomethane molecule transfers a methyl group to the oxygen atom of the carboxylic acid, while liberating nitrogen gas (a very stable product). WWU-Chemistry
Mechanism of Methyl Ester Formation This process is very favorable energetically, owing to the great stability of N2. WWU-Chemistry
Reactions of Esters WWU-Chemistry
By far the most important reaction of the esters is hydrolysis. Hydrolysis can take place in either acidic or basic solution. WWU-Chemistry
Hydrolysis of Esters WWU-Chemistry
Example Can anyone suggest why an ester might not make a suitable perfume? WWU-Chemistry
Another Example Notice that, in a basic medium, the product of the hydrolysis is a carboxylate ion. The basic hydrolysis of an ester is often known as a saponification. WWU-Chemistry
Hydrolysis of an Ester in Base WWU-Chemistry
Hydrolysis of an Ester in Acid WWU-Chemistry
Saponification – The World’s Second-Oldest Known Organic Reaction • Recall that the triacylglycerols (simple lipids or fats) are esters. WWU-Chemistry
If we hydrolyze a fat in aqueous base: The products are three equivalents of carboxylate ion. WWU-Chemistry
If our starting fat were something like tristearin, which would be representative of an animal fat: WWU-Chemistry
Then basic hydrolysis would give us three equivalents of the corresponding sodium carboxylate. This is the structural formula for ordinarysoap! (“Grandma’s lye soap”) “Saponification” (from Latin: sapo = soap). Saponification is a soap-making reaction. WWU-Chemistry
Transesterification • Sometimes, it is more convenient to convert the alkyl group of an ester to another alkyl group. • This process is known as transesterification. • Transesterification can take place under either acidic or basic conditions, although most commonly it is done under acidic conditions. WWU-Chemistry
Transesterification WWU-Chemistry
Transesterification (in base) WWU-Chemistry
Example Thus, we’ve converted a methyl ester to a benzyl ester. WWU-Chemistry
A Problem: Suppose we have a sample of a fat. WWU-Chemistry
What are R1, R2, and R3? • With what frequency do they appear in a sample of the fat? • What is their relative distribution in the sample? • We can’t analyze the fat sample directly, because it isn’t very soluble and it isn’t volatile. • We need to convert it from a glycerol ester to some more convenient form of ester. WWU-Chemistry
Transesterification WWU-Chemistry
The methyl esters are much more volatile. • The transesterified fat sample can now be analyzed by gas chromatography. WWU-Chemistry
Reaction of Esters with Organometallic Reagents WWU-Chemistry
We can outline (crudely) the mechanism as follows: WWU-Chemistry
BUT: The ketone can also react with the organometallic reagent! So, addition of organometallic reagent happens a second time. So, the product is a tertiary alcohol. WWU-Chemistry