HPG Axis
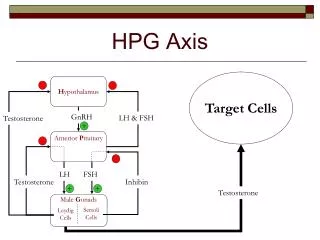
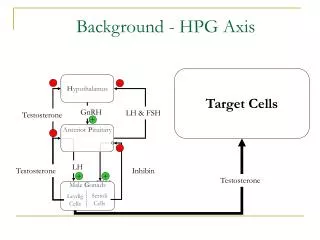
HPG Axis. Target Cells. H ypothalamus. -. -. GnRH. LH & FSH. Testosterone. +. Anterior P ituitary. -. -. LH. FSH. Testosterone. Inhibin. +. +. Testosterone. Male G onads. Sertoli Cells. Leydig Cells. Hypogonadism. Hypogonadism

HPG Axis
E N D
Presentation Transcript
HPG Axis Target Cells Hypothalamus - - GnRH LH & FSH Testosterone + Anterior Pituitary - - LH FSH Testosterone Inhibin + + Testosterone Male Gonads Sertoli Cells Leydig Cells
Hypogonadism • Hypogonadism • General: Reduction or loss of gonad function • Target function: Testosterone production by leydig cells found in male gonads • Approach: Restore steroidogenic function of leydig cells
Challenges with traditional cell transplantation Immune response Foreign body reaction Advantages of microencapsulation Cell entrapment Immunoisolation Selective transportation Sustained release of hormones from entrapped cells Reduced diffusion distance Cell Transplantation
Microcapsule Parameters Degradation Size exclusion via mesh size LH, FSH, O2, Nutrients Antibodies Testosterone, Wastes Biocompatibility Microcapsule Size
O PEGdA O O n O Polyethylene glycol (PEG) • Synthetic polymer • Systematically variable mesh size • Non-biodegradable • Sustained cell protection • Bio-inert • Difficult for cells & proteins to adhere Pre-cursor Solution: 10% PEGdA MW12000 0.05% I2959 PBS diluent ± cell suspension PEGdA macromers Photopolymerization (365nm UV light) Polymerization & cross-linking via free-radical mechanism Swelling PEGdA hydrogel H2O
Problem Design Statement To investigate the effects of hydrogel thickness on the viability of human prostate cancer cells embedded within a polyethylene glycol diacrylate hydrogel. Additionally, to assess the polymerization and cross-linking phenomena of PEGdA macromers and the diffusive behavior of progesterone through a PEGdA hydrogel matrix. The overall goal of this project is to design an encapsulation system that offers efficient immunoprotection and effective diffusion of oxygen, nutrients, hormones, and metabolic wastes. This system, along with embedded human prostate cancer cells, will enable the restoration of un-regulated hormone levels commonly observed in elders, and retard the symptoms of aging.
Previous Work • Used capsule size of 100µm diameter • Observed cell viability out to 7 days and detected negligible testosterone release • 15 min of UV exposure = threshold for sustained cell viability • Current approach for improvements • Microcapsule size • UV exposure time
UV Exposure Time vs. Degree of Hydrogel cross-linking 14.5 minutes of UV exposure is sufficient for cross linking 3D Swelling Ratio = 3.8
Thickness (µm) 0.0% 0 50 100 150 200 250 -10.0% -20.0% -30.0% Concentration Percent Change in Oxygen -40.0% -50.0% -60.0% Percent Change in Oxygen Concentration at Various Hydrogel Thicknesses as Compared to the Oxygen Concentration at the Site of Implantation Capsule Diameter • Post-Swell Testing Range = 25µm ~ 250µm
Tape spacers Microscope slides PEGdA Hydrogel Sigmacote Preset thickness Hydrogel sandwich • Simulation of capsule radius • Sigmacote surface treatment to aid PEGdA removal • Post-swell thickness = 25m ~ 250m • Pre-swell thickness = 25m ~ 175m
Ultrasound • Confirmation of swelling calculation • Determine pre/post swell thickness of hydrogel sandwich Transducer Water D PEGdA Microscope Slide Distance (D) = (1/2) x [Time x Speed of Sound]
Ultrasound Result • Linear swollen ratio is 1.54
Progesterone Diffusion • Observed Progesterone release over time • High progesterone levels after 5 hours • Progesterone level exceeded linear range of calibrated curve • Data variability • Sex hormone capable of diffusing out of PEGdA network
Cell Viability Results Statistical Analysis: 2-sample t-test α = 0.05 * * * * * * * Cell Titer-BlueTM Cell Viability Assay * Denotes significant drop from day 2 to day 3 * Denotes significant drop from day 3 to day 4
Cell Viability Discussion • Further data is needed to establish a meaningful trend and interpretation • Fluorescence readings close to that of the negative control (cell culture medium) • Increase number of cells per well and/or increase incubation time to 3 or 4 hours
Overall Conclusions • PEGdA suitable material for cell encapsulation • Sub-lethal UV time requirement @ 14.5 min • The mesh size achieved allows for the diffusion of progesterone • Need to extend cell viability studies for more concrete interpretation
Future Work • Continue to assess hydrogel thickness effect on cell viability (extended studies) • Evaluate effects of gel thickness on hormone release • 2-D & 3-D studies of the effects of RGD cell adhesion peptides on cell function • Fabrication of micro-spheres of specified diameter • In vivo analysis of encapsulation system
References • ALPCO Diagnostics (2004). Progesterone EIA: For the direct quantitative determination of Progesterone by enzyme Immunoassay in human serum.11-PROGH-305 Version 4.0 • Cruise, G. M., Hegre, O. D., Scharp, D. S., & Hubbell, J. A. (1998). A sensitivity study of the key parameters in the interfacial photopolymerization of poly(ethylene glycol) diacrylate upon porcine islets. Biotechnology and bioengineering, 57(6), 655-665. • Cruise, G. M., Scharp, D. S., & Hubbell, J. A. (1998). Characterization of permeability and network structure of interfacially photopolymerized poly(ethylene glycol) diacrylate hydrogels. Biomaterials, 19(14), 1287-1294. • Diramio, J. A., Kisaalita, W. S., Majetich, G. F., & Shimkus, J. M. (2005). Poly(ethylene glycol) methacrylate/dimethacrylate hydrogels for controlled release of hydrophobic drugs. Biotechnology progress, 21(4), 1281-1288. • Kizilel, S., Perez-Luna, V. H., & Teymour, F. (2004). Photopolymerization of poly(ethylene glycol) diacrylate on eosin- functionalized surfaces. Langmuir : the ACS journal of surfaces and colloids, 20(20), 8652-8658. • Kizilel, S., Sawardecker, E., Teymour, F., & Perez-Luna, V. H. (2006). Sequential formation of covalently bonded hydrogel multilayers through surface initiated photopolymerization. Biomaterials, 27(8), 1209-1215. • Martens, P. J., Bryant, S. J., & Anseth, K. S. (2003). Tailoring the degradation of hydrogels formed from multivinyl poly(ethylene glycol) and poly(vinyl alcohol) macromers for cartilage tissue engineering. Biomacromolecules, 4(2), 283-292. • Mellott. M, Searcy. K, Pishko. M (2001). Release of protein from highly cross-linked hydrogels of poly(ethylene glycol) diacrylate fabricated by UV polymerization. Biomaterials 22(9):929-41. • Muschler. G, Nakamoto C, Griffth L (2004). Engineering Principles of Clinical Cell-Based Tissue Engineering. The Journal of Bone and Joint Surgery (American) 86:1541-1558 • Nuttelman, C. R., Tripodi, M. C., & Anseth, K. S. (2005). Synthetic hydrogel niches that promote hMSC viability. Matrix biology : journal of the International Society for Matrix Biology, 24(3), 208-218. • Yang. F, Williams. C, Wang. D, Lee. H (2004) The effect of incorporating RGD adhesive peptide in polyethylene glycol diacrylate hydrogel on osteogenesis of bone marrow stromal cells. Biomaterials. 2005 Oct;26(30):5991-8.
Special Thanks • Chemistry Department • Dr. Daesung Lee • Yi-Jin Kim • VA hospital • Yi-Jin Kim • Dr. Craig Atwood • Miguel Gallego • Andrea Wilson • Ryan Haasl • Promega Corporation • Lydia Hwang for her vital donation of project resources • CS Hyde Company • School of Medicine and Public Health, Medical Physics • Dr. Tim Stiles for his help in ultrasound measurements • Pharmacy Department • Dr. John Kao • Graduate student Amy Chung for her endless generosity • Biomedical Engineering Department • Dr. Kristyn Masters and lab • Dr. William Murphy and lab • Dr. Brenda Ogle and lab
Micro Albert Kwansa Eric Lee
encaps John Harrison Miguel Benson Client: Dr. Craig Atwood Advisor: Professor William Murphy
ulation Yik Ning Wong