Understanding Perchlorates: Essential for Airbag Functionality
In airbag manufacturing, perchlorates are used for their powerful oxidising properties, facilitating the combustion of the propellant material. This reaction allows airbags to inflate within milliseconds, providing vital protection in the event of an accident.<br><br>Source - https://calibrechem.com/blog/perchlorates-in-the-manufacture-of-airbags-a-technical-perspective/

Understanding Perchlorates: Essential for Airbag Functionality
E N D
Presentation Transcript
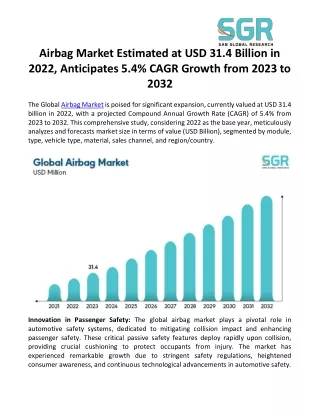
mail@calibrechem.com calsales@calibrechem.com 91 - 22 - 24330002 Select Language About Products Industries Capabilities Systems Contact Sustainability Perchlorates in the Manufacture of Airbags: A Technical Perspective Blog > Perchlorates in the Manufacture of Airbags: A Technical Perspective Perchlorates in the Manufacture of Airbags: A Technical Perspective The evolution of automobile safety has witnessed significant advancements over the decades, with airbags standing out as one of the most revolutionary developments. Designed to inflate in milliseconds during a collision, airbags provide a crucial cushioning effect, significantly reducing the risk of severe injury or death. At the core of this life- saving technology lies a series of complex chemical reactions, one of the most vital being the role of perchlorates. These compounds, often overshadowed by their more visible counterparts in the automotive industry, play an indispensable role in the precise and rapid deployment of airbags. This blog delves into the technical aspects of perchlorates in airbag manufacturing, exploring their function, benefits, and the underlying science that makes them so effective. The Science Behind Airbags Before diving into the specifics of perchlorates, it’s essential to understand how airbags function. An airbag system comprises several components: sensors, inflator, and the airbag cushion itself. When a collision is detected, sensors send an electrical signal to the inflator, triggering a rapid chemical reaction. This reaction produces a gas that inflates the airbag almost instantaneously. The entire process takes place in less time than it takes to blink an eye, and the choice of chemicals used is crucial to ensure this rapid response. The Role of Perchlorates Perchlorates, a group of salts derived from perchloric acid, are integral to the airbag inflator’s function. Common perchlorates used in this application include ammonium perchlorate (NH₄ClO₄) and potassium perchlorate (KClO₄). These compounds serve as oxidising agents, a critical role in the generation of the gas that inflates the airbag. In the context of airbags, the oxidising property of perchlorates is harnessed to facilitate the rapid decomposition of a fuel, typically sodium azide (NaN₃) in older systems or guanidine nitrate (CH₆N₄O₃) in more modern designs. The decomposition of these fuels produces a large volume of nitrogen gas (N₂), which then inflates the airbag. The reaction can be summarised as follows: 2NaN3 →3N2 +2Na This reaction occurs in the presence of a heat-producing oxidiser like a perchlorate, which ensures that the decomposition happens rapidly enough to inflate the airbag in time to protect the occupant during a crash. Why Perchlorates? 1. High Reactivity: Perchlorates are highly reactive and release oxygen readily. This characteristic is essential for the rapid combustion of the fuel in the inflator, ensuring that the gas generation is almost instantaneous. This high reactivity allows for the precise control needed to inflate the airbag in milliseconds. ?. Stable Storage: Despite their reactivity, perchlorates are relatively stable at ambient conditions. This stability is crucial for the safety and reliability of airbag systems, as the inflator must remain inert and inactive during the vehicle’s lifespan, only reacting when triggered by the crash sensors. 3. High Oxygen Content: Perchlorates possess a high oxygen content, which is vital for sustaining the rapid combustion of the fuel without requiring an external source of oxygen. This self-sufficiency makes them ideal for sealed environments like airbag inflators, where the reaction must occur independently of the external atmosphere. 4. Controlled Decomposition: The decomposition of perchlorates can be controlled by adjusting the temperature and pressure conditions within the inflator. This control is critical for ensuring that the airbag inflates with the right force and speed, avoiding either under-inflation, which would reduce effectiveness, or over-inflation, which could cause injury. The Evolution of Perchlorate Use in Airbags The use of perchlorates in airbags has evolved over the years as manufacturers sought to improve safety, performance, and environmental impact. Early airbag designs relied heavily on sodium azide as the primary fuel, with perchlorates acting as the oxidiser. However, concerns about the toxicity and environmental impact of sodium azide led to the development of alternative fuels like guanidine nitrate. Modern airbag systems utilise a combination of guanidine nitrate and potassium perchlorate, which provides a safer and more environmentally friendly alternative. This shift has not only improved the safety profile of airbags but also enhanced their performance, as guanidine nitrate decomposes at a lower temperature than sodium azide, resulting in a more controlled inflation. Challenges and Considerations While perchlorates offer numerous benefits in airbag manufacturing, they are not without challenges. The primary concern with perchlorates is their environmental impact. Perchlorate compounds are highly soluble in water and can persist in the environment, posing risks to human health and ecosystems. This persistence has led to regulatory scrutiny and a push towards developing alternative oxidisers that are more environmentally benign. Additionally, the handling and storage of perchlorates require stringent safety measures due to their reactive nature. Manufacturers must ensure that the compounds are stored in a stable, dry environment to prevent accidental reactions that could lead to hazardous situations. Future Direction The automotive industry is constantly evolving, and with it, the technology and materials used in safety systems like airbags. Researchers are exploring alternative oxidisers that can match or exceed the performance of perchlorates while minimising environmental impact. Some of the potential alternatives being studied include metal nitrates and organic peroxides, which offer similar reactivity with a lower environmental footprint. In parallel, advancements in airbag design and deployment mechanisms are reducing the reliance on chemical inflators altogether. New technologies like compressed gas inflators and hybrid systems that combine chemical and compressed gas reactions are being developed. These systems promise to deliver the same level of protection with reduced chemical usage and improved environmental performance. Conclusion As a leading speciality chemical manufacturer, Calibre Chemicals recognises the importance of safety and sustainability in the automotive industry. Stringent quality control measures and a dedication to innovation back our commitment to providing high-quality perchlorates for use in airbag systems. We understand the critical role that perchlorates play in ensuring the rapid and reliable deployment of airbags, and we strive to support our customers with products that meet the highest safety and performance standards. Whether it’s improving existing products or developing new, eco-friendly alternatives, Calibre Chemicals is dedicated to driving progress in the speciality chemicals industry. References https://www.sciencedirect.com/topics/medicine-and-dentistry/perchlorate https://www.enviro.wiki/index.php?title=Perchlorate https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2681191/ https://patents.google.com/patent/US7147733B2/en https://en.wikipedia.org/wiki/Airbag Privacy Policy Disclaimer Copyright © 2025 Calibre Chemicals Pvt. Ltd. Designed & Managed by Vizcom Solutions