Gene Expression
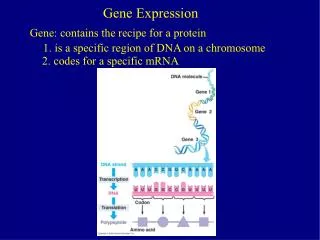
Gene Expression. Review covering RNA Processing & Translation. Posttranscriptional ( RNA Processing). - Transcription has created a primary transcript (pre mRNA) that is not ready to be exported from the nucleus to the ribosome in the cytosol.

Gene Expression
E N D
Presentation Transcript
Gene Expression Review covering RNA Processing & Translation
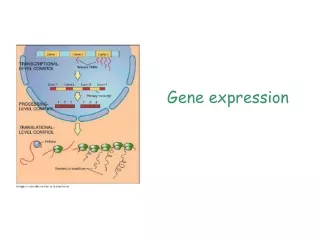
Posttranscriptional (RNA Processing) - Transcription has created a primary transcript (pre mRNA) that is not ready to be exported from the nucleus to the ribosome in the cytosol. • In order to be “competent for translation”, the pre mRNA must first be: • 1. Capped • 2. Tailed • 3. Spliced ** Not that in prokaryotic cells, mRNA transcripts are translated without these types of manipulations to the nucleic acid, this is evidenced by the fact that translation of the mRNA often begins before transcription has finished. Additionally, prokaryotic cells do not have a nucleus and so gene expression occurs in the cytoplasm.
5’ Capping = The Addition of a 7-methylguanosine Cap • This functions to: • 1. Protect the mRNA from nucleolytic degradation. • 2. Create a 5’-5’ triphosphate bridge. • 3. Assist the ribosome in recognizing the start of translation. • This involves several enzymatic reactions: • RNA Triphosphatase- Cleaves the leading phosphate group from the pre RNAs 5’ terminal triphosphate group • Capping Enzyme- Guanylates the pre mRNA, using GTP. This creates the 5’-5’ triphosphate bridge. • 7-guanosine-methyltransferase- Methylates the mRNA, using S-adenosylmethionine (SAM) to provide the –CH3 group. • 2’-O-methyltransferase- Methylates the 1st and 2nd nucleotides of the pre mRNA • Requires SAM
Mature eukaryotic mRNAs have well defined 3’ ends terminating in Poly (A) tails: • Polyadenylated tail is around 250 nucleotides long that consists of a highly conserved AAUAAA. • Functions to: • Protect the mRNA • Label the mRNA for nuclear export and ribosomal recognition. ** Not present in histones - Enzymatically added to the pre mRNA in two reactions: 1. Cleavage- Occurs around 20 nucleotides past the AAUAAA sequence, and less than 50 nucleotides before the U or GU rich sequence. • This reaction eliminates the need for perfect termination of transcription 2. Poly (A) polymerase (PAP)- Creates the poly (A) tail, using ATP • PAP is a template-independent RNAP that lengthens any mRNA primer that has a free 3’ hydroxyl group. • PAP is activated by cleavage and polyadenylation specifity factor (CPSF) ,which recognizes the AAUAAA sequence and then disassociates; much like the sigma factor in prokaryotes. • CPSF binds to RNAP IIs phosphorylated C terminal domain. ** Deleting CPSF will INHIBIT polyadenylation** • ** Please note: The poly (A) tail is not needed for mRNA translation; however it functions to protect the mRNA in the cytosol. This is evidenced by the fact that histones are the only mature mRNAs that lack AUAAA sequences**
Splicing • Primary transcripts are also called (pre mRNAs) • This mRNA includes introns, which are nonexpressed and constitute much of the genome. • Capping occurs after initiation, with splicing commencing during the elongation phase of transcription. • mRNA that is ready for translation (mature RNA) after splicing is complete and the RNA has been polyadenylated; and at this point it is exported to the cytosol where ribosomes use it to create protein.
Splicing occurs in a two stage transesterification reaction: 1. Lariat Structure- is formed when a 2’,5’ phosphodiester bond is created between an intron adenosine residue and the intron’s 5’ terminal phosphate group. • At this point the 5’ exon is released, creating the loop (lariat) * Mutating a branch point adenosine residue destroys the ability to splice at this site. • 2.Spliced product- Created when 5’ exons free 3’ –OH group displaces the 3’ end of the intron. This forms a phosphodiester bond with the 5’ terminal phosphate of the exon and the 3’.The loop is later degraded. *Mutations that change the AG at the 3’ splice junction block this step, but do not block loop formation. ** These two transesterification reactions do not require energy input, as the energy is transferred through the concomitant formation of new phosphodiester bonds.
Details of Splicing • U1 snRNP “Snurp”- recognizes and cleaves 5’ of intron. • U2 snRNP- recognizes the branch point adenine. • Spliceosome (45S) Complex- splices 5’ end to branch point adenine • Small nuclear RNAs (snRNAs)- form complexes to make snRNPS • U6- catalytically essential metal ion that enhances nucleophilicity of OH and stabilizes the leaving group in the reaction. ** intron removal occurs before transcription of pre RNA is complete.**
Translation tRNAs (Transfer RNA)- Carry amino acids to be used in protein synthesis to the Ribosome. tRNA Structure
tRNA Structure: • tRNAs can be schematically arranged into a cloverleaf secondary structure and have 6 common features: 1. a 5’-terminal phosphate group 2. Acceptor stem- The amino acid residue carried by the tRNA is covalently attached to its 3’-terminal OH group. 3. D arm- D for dihydrouridine, which is part of the loop of the arm. 4. Anticodon arm- contains the anticodon sequence. The 3’ purine is invariable modified. 5. TψC or T arm- pseudouridine 6. CCA sequence- 3’ sequence with a free OH group. May be genetically specified of enzymatically added to immature tRNA.
tRNA Structure 2 • tRNA also has: • 15 invariant positions that do not change, and 8 semi-invariant positions- that are only purine/ only pyrimidine which occur mostly in the loop regions. • Variable arm- site of greatest variability. • None of these modifications are essential for maintaining a tRNA’s structural integrity or for its proper binding to the ribosome. However, base modifications may help promote attachment of the proper amino acid to the acceptor stem or strengthen codon–anticodon interactions.
Enzymatic reactions allow the tRNA to become charged so it can carry the amino acid to the ribosome: • Aminoacyl-tRNA synthetase-produces the charged amino acid. • Amino acid activation- • 1. AA + ATP AA-AMP + pyrophosphate (2Pi) • 2. AA-AMP + tRNA AA-TRNA + AMP • * The aa-tRNA (aminoacyl-adenylate) is a high energy compound. • In all but three aaRSs, aminoacyl-adenylate creation can occur in the absence of tRNAs. • The overall reaction is driven to completion by the hydrolysis of the PPi generated in the first step.
Ribosomes: • The functions of the ribosome: • The complexity of ribosomes allows it to carry out the following vital functions: • The ribosome binds mRNA such that its codons can be read with high fidelity. • The ribosome includes specific binding sites for tRNA molecules. • The ribosome mediates the interactions of nonribosomal protein factors that promote polypeptide chain initiation, elongation, and termination. • The ribosome catalyzes peptide bond formation. • The ribosome undergoes movement so that it can translate sequential codons.
The Prokaryotic Ribosome • Has a sedimentation coefficient of 70S. • The ribosome can be dissociated into two unequal subunits. • The small (30S) subunit consists of a 16S rRNA molecule and 21 different proteins. • The large (50S) subunit contains a 5S and a 23S rRNA together with 31 different proteins. • Ribosomes have three functionally distinct tRNA-binding sites: • The A or aminoacyl site (it accommodates the incoming aminoacyl–tRNA) • The P or peptidyl site (it accommodates the peptidyl–tRNA, the tRNA to which the growing peptide chain is attached) • The E or exit site (it accommodates a deacylated tRNA that is about to exit the ribosome).
The Eukaryotic Ribosome: • Although eukaryotic and prokaryotic ribosomes resemble each other in both structure and function, they differ in nearly all details. • A nominal sedimentation coefficient of 80S. • They dissociate into two unequal subunits with compositions that are distinctly different from those of prokaryotes. • The small (40S) consists of 33 unique polypeptides and an 18S rRNA. • Its large (60S) subunit contains 49 different polypeptides and three rRNAs of 28S, 5.8S, and 5S. • The additional complexity of the eukaryotic ribosome relative to its prokaryotic counterpart is presumably due to the eukaryotic ribosome’s additional functions: • Its mechanism of translational initiation is more complex; it must be transported from the nucleus, where it is formed, to the cytoplasm, where translation occurs; and the machinery with which it participates in the secretory pathway is more complicated.
Translation • Ribosomes orchestrate the translation of mRNA to synthesize polypeptides: • Polypeptide synthesis proceeds from the N-terminus to the C-terminus. • Chain elongation occurs by linking the growing polypeptide to the incoming tRNA’s amino acid residue • Ribosomes read mRNA in the 5′ → 3′ direction. • Because mRNA is also synthesized in the 5′ → 3′ direction, prokaryotic ribosomes can commence translation as soon as a nascent mRNA emerges from RNA polymerase. This, however, is not possible in eukaryotes because the nuclear membrane separates the site of transcription (the nucleus) from the site of translation (the cytosol). • Active translation occurs on polysomes
The Mechanism: • Initiation: • IF-2 + GTP bind to the start codon (AUG/Met) • Small subunit find the kozac sequence (ACCAUGG) • Shine-Dalgarno in prokaryotes (AGGAGG) • Initiation begins at AUG • Initiation Factors: • IF-1: Assists IF-3 • IF-2:Binds initiator tRNA to GTP • IF-3: Releases mRNA and tRNA from subunit
The Mechanism: • Elongation: (Decoding->Transpeptidation->Translocation) • -tRNA binds to the A-site • Requires EF-Tu, EF-Ts and GTP • Peptidyl Transferase of large subunit forms peptide bond is without input of ATP/GTP (Ribosomes are ribozymes) • mRNA translocates 5’ to 3’ to next three codons • Peptidyl moves from A site to P site • tRNA moves from the P site to the E site, driven by EF-G and GTP • Elongation Factors: • EF-Tu:Binds aa-tRNA to GTP • EF-Ts: Displaces GDP from EF-Tu • EF-G:Promotes translocation through GTP binding and hydrolysis.
The Mechanism: • Termination: • RFs mimic tRNAs and bind to stop codons • Use GTP to bind the protein to water, terminating the chain. • Release Factors • RF-1: Recognizes UAA and UAG stops • RF-2: Recognizes UAA and UGA stops • RF-3: Stimulates RF-1 and 2 release via GTP hydrolysis • RRF:Together with EF-G, induces ribosomal dissociation of the small and large subunits.
Summary: 1. The primary transcripts of most eukaryotic structural genes are posttranscriptionally modified by the addition of a 5′ cap and a 3′ poly(A) tail. mRNAs that contain introns undergo splicing, in which the introns are excised and the exons are joined together via two transesterification reactions mediated by an snRNA-containing spliceosome. 2. The processing of pre-rRNAs includes nucleolytic cleavage and snoRNA-assisted methylation. Some eukaryotic rRNA transcripts undergo splicing catalyzed by the intron itself. 3. tRNA transcripts may be processed by the addition, removal, and modification of nucleotides. 4. The genetic code, by which nucleic acid sequences are translated into amino acid sequences, is composed of three-nucleotide codons that do not overlap and are read sequentially by the protein-synthesizing machinery. 5. The standard genetic code of 64 codons includes numerous synonyms, three Stop codons, and one initiation codon. 6. All tRNAs have numerous chemically modified bases and a similar cloverleaf secondary structure comprising an acceptor stem, D arm, TψC arm, anticodon arm, and variable arm. The three-dimensional structures of tRNAs are likewise similar and are maintained by stacking interactions and non-Watson–Crick hydrogen-bonded cross-links. 7. Aminoacyl–tRNA synthetases (aaRSs) catalyze the ATP-dependent attachment of an amino acid to the appropriate tRNA to yield an aminoacyl–tRNA (aa–tRNA). The acceptor stem and anticodon loop are common identity elements for tRNA–aaRS interactions. The fidelity of aminoacylation is enhanced by proofreading.
Summary: 8. Wobble pairing between mRNA codons and tRNA anticodons at the third position accounts for much of the degeneracy of the genetic code. 9. Ribosomes, which are large complexes of RNA and protein, have a structure that is determined by their RNA components. A small subunit and large subunit associate to form the intact ribosome, which accommodates an aa–tRNA in the A site, a peptidyl–tRNA in the P site, and a deacylated tRNA in the E site. 10. During translation initiation, an initiator tRNA charged with fMet (prokaryotes) or Met (eukaryotes), an mRNA with an AUG initiation codon, and the ribosomal subunits assemble. Initiation requires GTP-hydrolyzing initiation factors. The initiating AUG codon is identified in prokaryotes via the mRNA’s Shine–Dalgarno sequence. In eukaryotes it is the mRNA’s initiating AUG that is identified in a complex process by its proximity to the mRNA’s 5′ cap. 11. A polypeptide chain is elongated from its N- to its C-terminus. An aa–tRNA in complex with a GTP-hydrolyzing elongation factor binds to the A site, where the ribosome senses correct codon–anticodon pairing, a process whose accuracy is enhanced by proofreading. The peptidyl transferase activity of the large subunit’s RNA catalyzes the attack of the A-site bound aa–tRNA’s amino group on the peptidyl–tRNA in the P site. Following transpeptidation, a second GTP-hydrolyzing elongation factor promotes the translocation of the new peptidyl–tRNA to the P site. 12.Translation termination requires release factors that recognize Stop codons.