The Mole Capacitor
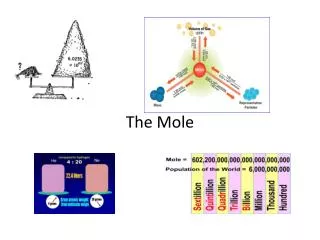
The Mole Capacitor. Notes and Practice Problems. x 22.4 L/mole . x 6.02 x 10 23 particles/mole. ÷ 6.02 x 10 23 particles/mole . ÷ 22.4 L/mole. ÷ molar mass (g/mole) . x molar mass (g/mole). x 22.4 L/mole .


The Mole Capacitor
E N D
Presentation Transcript
The Mole Capacitor Notes and Practice Problems
x 22.4 L/mole x 6.02 x 1023 particles/mole ÷ 6.02 x 1023 particles/mole ÷ 22.4 L/mole ÷molar mass (g/mole) x molar mass (g/mole)
x 22.4 L/mole x 6.02 x 1023 particles/mole ÷ 6.02 x 1023 particles/mole ÷ 22.4 L/mole ÷molar mass (g/mole) x molar mass (g/mole)
“Chemical Battlefield” • Crystals of potassium permanganate (KMnO4) will be added to a test tube that contains separate layers of concentrated sulfuric acid (H2SO4) and methanol (CH3OH). As the potassium permanganate crystals settle upon the sulfuric acid/methanol interface, they oxidize and result in several small, bright explosions. • If 3.57 x 1022 molecules of potassium permanganate are used, how many moles of potassium permanganate is this? • 3.57 x 1022 molecules • 6.02 x 1023 molecules/mole • = 0.0593 moles
“Chemical Battlefield” • Crystals of potassium permanganate (KMnO4) will be added to a test tube that contains separate layers of concentrated sulfuric acid (H2SO4) and methanol (CH3OH). As the potassium permanganate crystals settle upon the sulfuric acid/methanol interface, they oxidize and result in several small, bright explosions. • Assuming that 0.125 moles of methanol are used, how many grams is this? • 0.125 moles • = 4.00 g x g/mole 32.0 C 1 x 12.0 = 12.0 H 4 x 1.0 = 4.0 O 1 x 16.0 =16.0 molar mass = 32.0 g/mole
“Chemical Battlefield” • Crystals of potassium permanganate (KMnO4) will be added to a test tube that contains separate layers of concentrated sulfuric acid (H2SO4) and methanol (CH3OH). As the potassium permanganate crystals settle upon the sulfuric acid/methanol interface, they oxidize and result in several small, bright explosions. • If 0.507 moles of sulfuric acid are used, how many molecules would this be?
“Chemical Battlefield” • Crystals of potassium permanganate (KMnO4) will be added to a test tube that contains separate layers of concentrated sulfuric acid (H2SO4) and methanol (CH3OH). As the potassium permanganate crystals settle upon the sulfuric acid/methanol interface, they oxidize and result in several small, bright explosions. • If 2.13 g of potassium permanganate is used, how many moles is this?
“The Tower” • A 40 mL portion of concentrated sulfuric acid (H2SO4) will be added to a beaker of sugar (sucrose, C12H22O11). As the sugar oxidizes, several gases are produced, including carbon dioxide and water vapor. These hot gases expand and push the oxidized sugar out of the beaker. • If the sample of sulfuric acid contains 0.240 moles, how many grams of sulfuric acid are there? • 0.240 moles • 82.1 g C 12 x 12.0 = 144.0 x g/mole 342.0 H 22 x 1.0 = 22.0 O 11 x 16.0 =176.0 molar mass = 342.0 g/mole
“The Tower” • A 40 mL portion of concentrated sulfuric acid (H2SO4) will be added to a beaker of sugar (sucrose, C12H22O11). As the sugar oxidizes, several gases are produced, including carbon dioxide and water vapor. These hot gases expand and push the oxidized sugar out of the beaker. • If 12.2 L of carbon dioxide gas are produced, how many moles is this? • 12.2 L • 22.4 L/mole • 0.545 moles
“The Tower” • A 40 mL portion of concentrated sulfuric acid (H2SO4) will be added to a beaker of sugar (sucrose, C12H22O11). As the sugar oxidizes, several gases are produced, including carbon dioxide and water vapor. These hot gases expand and push the oxidized sugar out of the beaker. • If 36.9 g of sugar are used, how many moles is this?
“The Tower” • A 40 mL portion of concentrated sulfuric acid (H2SO4) will be added to a beaker of sugar (sucrose, C12H22O11). As the sugar oxidizes, several gases are produced, including carbon dioxide and water vapor. These hot gases expand and push the oxidized sugar out of the beaker. • If the reaction also produced 1.38 moles of water vapor, how many liters would this be?