Protein Methods
Protein Methods. Andy Howard Introductory Biochemistry Fall 2010, IIT. Proteins are worth studying. We’ll perform a quick overview of methods of studying proteins Purification methods Analytical methods Structural methods. The Protein Data Bank. http://www.rcsb.org/

Protein Methods
E N D
Presentation Transcript
Protein Methods Andy Howard Introductory BiochemistryFall 2010, IIT
Proteins are worth studying • We’ll perform a quick overview of methods of studying proteins • Purification methods • Analytical methods • Structural methods Protein Methods and Function
The Protein Data Bank • http://www.rcsb.org/ • This is an electronic repository for three-dimensional structural information of polypeptides and polynucleotides Protein Methods and Function
What it contains • 68000 structures as of September 2010 • Most are determined by X-ray crystallography • Smaller number are high-field NMR structures • A few calculated structures, most of which are either close relatives of experimental structures or else they’re small, all-alpha-helical proteins Protein Methods and Function
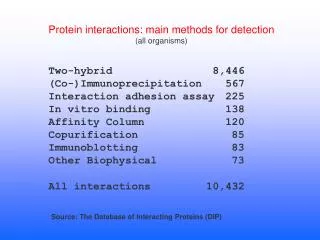
What you can do with the PDB • Display structures • Look up specific coordinates • Run clever software that compares and synthesizes the knowledge contained there • Use it as a source for determining additional structures Protein Methods and Function
Protein Purification • Why do we purify proteins? • To get a basic idea of function we need to see a protein in isolation from its environment • That necessitates purification • An instance of reductionist science • Full characterization requires a knowledge of the protein’s action in context Protein Methods and Function
Salting Out • Most proteins are less soluble in high salt than in low salt • In high salt, water molecules are too busy interacting with the primary solute (salt) to pay much attention to the secondary solute (protein) • Various proteins differ in the degree to which their solubility disappears as [salt] goes up • We can separate proteins by their differential solubility in high salt. Protein Methods and Function
How to do it • Dissolve protein mixture in highly soluble salt like Li2SO4, (NH4)2SO4, NaCl • Increase [salt] until some proteins precipitate and others don’t • You may be able to recover both: • The supernatant (get rid of salt; move on) • The pellet (redissolve, desalt, move on) • Typical salt concentrations > 1M Protein Methods and Function
Dialysis • Some plastics allow molecules to pass through if and only ifMW < Cutoff • Protein will stayinside bag, smaller proteins will leave • Non-protein impurities may leave too. Protein Methods and Function
Gel-filtration chromatography • Pass a protein solution through a bead-containing medium at low pressure • Beads retard small molecules • Beads don’t retard bigger molecules • Can be used to separate proteins of significantly different sizes • Suitable for preparative work Protein Methods and Function
Ion-exchange chromatography • Charged species affixed to column • Phosphonates (-) retard (+)charged proteins:Cation exchange • Quaternary ammonium salts (+) retard (-)charged proteins:Anion exchange • Separations facilitated by adjusting pH Protein Methods and Function
Affinity chromatography • Stationary phase contains a species that has specific favorable interaction with the protein we want • DNA-binding protein specific to AGCATGCT: bind AGCATGCT to a column, and the protein we want will stick; every other protein falls through • Often used to purify antibodies by binding the antigen to the column Protein Methods and Function
Metal-ion affinity chromatography • Immobilize a metal ion, e.g. Ni, to the column material • Proteins with affinity to that metal will stick • Wash them off afterward with a ligand with an even higher affinity • We can engineer proteins to contain the affinity tag:poly-histidine at N- or C-terminus Protein Methods and Function
High-performance liquid chromatography • Many LC separations can happen faster and more effectively under high pressure • Works for small molecules • Protein application is routine too, both for analysis and purification • FPLC is a trademark, but it’s used generically Protein Methods and Function
Electrophoresis • Separating analytes by charge by subjecting a mixture to a strong electric field • Gel electrophoresis: field applied to a semisolid matrix • Can be used for charge (directly) or size (indirectly) Protein Methods and Function
SDS-PAGE • Sodium dodecyl sulfate: strong detergent, applied to protein • Charged species binds quantitatively • Denatures protein • Good because initial shape irrelevant • Bad because it’s no longer folded • Larger proteins move slower because they get tangled in the matrix • 1/Velocity √MW Protein Methods and Function
SDS PAGE illustrated Protein Methods and Function
Isoelectric focusing I • Protein applied to gel without charged denaturant • Electric field set up over a pH gradient (typically pH 2 to 12) • Protein will travel until it reaches the pH where charge =0 (isoelectric point) Protein Methods and Function
Isoelectric focusing II • Sensitive to single changes in charge (e.g. asp -> asn) • Can be readily used preparatively with samples that are already semi-pure Protein Methods and Function
Ultraviolet spectroscopy • Tyr, trp absorb and fluoresce:abs ~ 280-274 nm; f = 348 (trp), 303nm (tyr) • Reliable enough to use for estimating protein concentration via Beer’s law • UV absorption peaks for cofactors in various states are well-understood • More relevant for identification of moieties than for structure determination • Quenching of fluorescence sometimes provides structural information Protein Methods and Function
Warning: Specialty Content! • I determine protein structures (and develop methods for determining protein structures) as my own research focus • So it’s hard for me to avoid putting a lot of emphasis on this material • But today I’m allowed to do that, because it’s one of the stated topics of the day. Protein Methods and Function
How do we determine structure? • We can distinguish between methods that require little prior knowledge (crystallography, NMR, ?CryoEM?)and methods that answer specific questions (XAFS, fiber, …) • This distinction isn’t entirely clear-cut Protein Methods and Function
Crystallography: overview • Crystals are translationally ordered 3-D arrays of molecules • Conventional solids are usually crystals • Proteins have to be coerced into crystallizing • … but once they’re crystals, they behave like other crystals, mostly Protein Methods and Function
How are protein crystals unusual? • Aqueous interactions required for crystal integrity: they disintegrate if dried • Bigger unit cells (~10nm, not 1nm) • Small # of unit cells and static disorder means they don’t scatter terribly well • So using them to determine 3D structures is feasible but difficult Protein Methods and Function
Crystal structures: Fourier transforms of diffraction results • Experiment: • Grow crystal, expose it to X-ray • Record diffraction spots • Rotate through small angle and repeat ~180 times • Position of spots tells you size, shape of unit cell • Intensity tells you what the contents are • We’re using electromagnetic radiation, which behaves like a wave, exp(2ik•x) • Therefore intensity Ihkl = C*|Fhkl|2 Protein Methods and Function
What are these Fhkl values? • Fhkl is a complex coefficient in the Fourier transform of the electron density in the unit cell:(r) = (1/V) hklFhkl exp(-2ih•r) • Critical point: any single diffraction spot contains information derived from all the atoms in the structure; and any atom contributes to all the diffraction spots Protein Methods and Function
The phase problem Fhkl • Note that we saidIhkl = C*|Fhkl|2 • That means we can figure out|Fhkl| = (1/C)√Ihkl • We can’t figure out the direction of F:Fhkl = ahkl + ibhkl = |Fhkl|exp(ihkl) • This direction angle is called a phase angle • Because we can’t get it from Ihkl, we have a problem: it’s the phase problem! bhkl ahkl Protein Methods and Function
What can we learn? • Electron density map + sequence we can determine the positions of all the non-H atoms in the protein—maybe! • Best resolution possible: Dmin = / 2 • Often the crystal doesn’t diffract that well, so Dmin is larger—1.5Å, 2.5Å, worse • Dmin ~ 2.5Å tells us where backbone and most side-chain atoms are • Dmin ~ 1.2Å: all protein non-H atoms, most solvent, some disordered atoms; some H’s Protein Methods and Function
What does this look like? • Takes some experience to interpret • Automated fitting programs work pretty well with Dmin < 2.1Å ATP binding to a protein of unknown function: S.H.Kim Protein Methods and Function
How’s the field changing? • 1990: all structures done by professionals • Now: many biochemists and molecular biologists are launching their own structure projects as part of broader functional studies • Fearless prediction: by 2020: • crystallographers will be either technicians or methods developers • Most structures will be determined by cell biologists & molecular biologists Protein Methods and Function
Macromolecular NMR • NMR is a mature field • Depends on resonant interaction between EM fields and unpaired nucleons (1H, 15N, 31S) • Raw data yield interatomic distances • Conventional spectra of proteins are too muddy to interpret • Multi-dimensional (2-4D) techniques:initial resonances coupled with additional ones Protein Methods and Function
Typical protein 2-D spectrum • Challenge: identify whichH-H distance is responsible for a particular peak • Enormous amount of hypothesis testing required Prof. Mark Searle,University of Nottingham Protein Methods and Function
Results of NMR studies • Often there’s a family of structures that satisfy the NMR data equally well • Can be portrayed as a series of threads tied down at unambiguous assignments • They portray the protein’s structure in solution • The ambiguities partly represent real molecular diversity; but they also represent atoms that area in truth well-defined, but the NMR data don’t provide the unambiguous assignment Protein Methods and Function
Comparing NMR to X-ray • NMR family of structures often reflects real conformational heterogeneity • Nonetheless, it’s hard to visualize what’s happening at the active site at any instant • Hydrogens sometimes well-located in NMR;they’re often the least defined atoms in an X-ray structure • The NMR structure is obtained in solution! • Hard to make NMR work if MW > 35 kDa Protein Methods and Function
What does it mean when NMR and X-ray structures differ? • Lattice forces may have tied down or moved surface amino acids in X-ray structure • NMR may have errors in it • X-ray may have errors in it (measurable) • X-ray structure often closer to true atomic resolution • X-ray structure has built-in reliability checks Protein Methods and Function
Cryoelectron microscopy • Like X-ray crystallography,EM damages the samples • Samples analyzed < 100Ksurvive better • 2-D arrays of molecules • Spatial averaging to improve resolution • Discerning details ~ 4Å resolution • Can be used with crystallography Protein Methods and Function
Circular dichroism • Proteins in solution can rotate polarized light • Amount of rotation varies with • Effect depends on interaction with secondary structure elements, esp. • Presence of characteristic patterns in presence of other stuff enables estimate of helical content Protein Methods and Function