Understanding Isotopes and Atomic Structure in Chemistry
Learn about isotopes, atomic number, atomic mass, protons, neutrons, electrons, and the importance of isotopes in elements. Explore examples and explanations in this comprehensive review of atomic structure.

Understanding Isotopes and Atomic Structure in Chemistry
E N D
Presentation Transcript
Review • Atomic Number = ______________
Review • Atomic Number = # protons • Atomic Mass = _______________
Review • Atomic Number = # protons • Atomic Mass = # protons + # neutrons • Neutrons = __________________
Review • Atomic Number = # protons • Atomic Mass = # protons + # neutrons • Neutrons = Atomic Mass – # protons
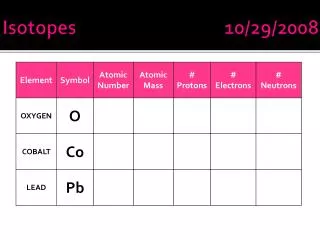
Isotopes 10/29/08 Review Atomic Number = Protons Atomic Mass = Protons + Neutrons Neutrons = Atomic Mass – Protons • In an uncharged atom, the number of protons is the same as the number of ____________. • electrons • An uncharged atom has NO charge. Why? • Protons = electrons
Isotopes What is the difference between these 2 atoms? Are they the same element?
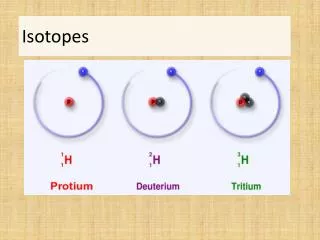
Isotopes • There can be different types (varieties) of an element • An isotope is a different form of an element but with a different number of neutrons • Same # of protons (same atomic number)
Isotopes • If the number of neutrons is different, what else is different about the element? • Atomic Mass = protons + neutrons • If the number of neutrons is different, the atomic mass is also different
Isotopes 10/29/08 Review Atomic Number = Protons Atomic Mass = Protons + Neutrons Neutrons = Atomic Mass – Protons Isotopes Different forms of an element - Element with different # of neutrons - Atomic number is still the same • In an uncharged atom, the number of protons is the same as the number of ____________. • electrons • An uncharged atom has NO charge. Why? • Protons = electrons
Isotopes • Not all elements have isotopes • If isotopes exist, the most common form of the element is the one found on the periodic table
Isotopes 10/29/08 Review Atomic Number = Protons Atomic Mass = Protons + Neutrons Neutrons = Atomic Mass – Protons Isotopes Different forms of an element - Element with different # of neutrons - Atomic number is still the same - # of proton is the same - Atomic Mass is different - most common form found on periodic table • In an uncharged atom, the number of protons is the same as the number of ____________. • electrons • An uncharged atom has NO charge. Why? • Protons = electrons
Isotope Symbols • Isotopes are abbreviated with the element and the atomic mass • Example: Helium-3 (He-3) the atomic mass is 3 how many neutrons does it have? Atomic mass – protons 3 – 2 = 1
Isotopes 10/29/08 Representing Isotopes Element + Atomic Mass EX. Helium-3 • In an uncharged atom, the number of protons is the same as the number of ____________. • electrons • An uncharged atom has NO charge. Why? • Protons = electrons
Isotopes 10/29/08 Representing Isotopes Element + Atomic Mass EX. Helium-3 Examples EX 1: Hawkium-10 (Hk) has an atomic number of 5 and a mass number of 10. • # protons: • In an uncharged atom, the number of protons is the same as the number of ____________. • electrons • An uncharged atom has NO charge. Why? • Protons = electrons
Isotopes 10/29/08 Representing Isotopes Element + Atomic Mass EX. Helium-3 Examples EX 1: Hawkium-10 (Hk) has an atomic number of 5 and a mass number of 10. • # protons: 5 • # electrons: • In an uncharged atom, the number of protons is the same as the number of ____________. • electrons • An uncharged atom has NO charge. Why? • Protons = electrons
Isotopes 10/29/08 Representing Isotopes Element + Atomic Mass EX. Helium-3 Examples EX 1: Hawkium-10 (Hk) has an atomic number of 5 and a mass number of 10. • # protons: 5 • # electrons: 5 • # neutrons: • In an uncharged atom, the number of protons is the same as the number of ____________. • electrons • An uncharged atom has NO charge. Why? • Protons = electrons
Isotopes 10/29/08 Representing Isotopes Element + Atomic Mass EX. Helium-3 Examples EX 1: Hawkium-10 (Hk) has an atomic number of 5 and a mass number of 10. • # protons: 5 • # electrons: 5 • # neutrons: 5 • In an uncharged atom, the number of protons is the same as the number of ____________. • electrons • An uncharged atom has NO charge. Why? • Protons = electrons
Isotopes 10/29/08 Examples EX 2: Hawkium-12 (Hk) has an atomic number of 5 and a mass number of 12. • # protons: • # electrons: • # neutrons: • In an uncharged atom, the number of protons is the same as the number of ____________. • electrons • An uncharged atom has NO charge. Why? • Protons = electrons
Isotopes 10/29/08 Examples EX 2: Hawkium-12 (Hk) has an atomic number of 5 and a mass number of 12. • # protons: 5 • # electrons: • # neutrons: • In an uncharged atom, the number of protons is the same as the number of ____________. • electrons • An uncharged atom has NO charge. Why? • Protons = electrons
Isotopes 10/29/08 Examples EX 2: Hawkium-12 (Hk) has an atomic number of 5 and a mass number of 12. • # protons: 5 • # electrons: 5 • # neutrons: • In an uncharged atom, the number of protons is the same as the number of ____________. • electrons • An uncharged atom has NO charge. Why? • Protons = electrons
Isotopes 10/29/08 Examples EX 2: Hawkium-12 (Hk) has an atomic number of 5 and a mass number of 12. • # protons: 5 • # electrons: 5 • # neutrons: 7 How do we know that Hawkium-12 and Hawkium-10 are isotopes? Hawkium-12 and Hawkium-10 are isotopes - same atomic number - different atomic mass different # neutrons • In an uncharged atom, the number of protons is the same as the number of ____________. • electrons • An uncharged atom has NO charge. Why? • Protons = electrons
PRACTICE • Find the following information for Nitrogen-13, Nitrogen-14, and Nitrogen-15 • Atomic Number: • Atomic Mass: • # Protons: • # Electrons: • # Neutrons: • Which isotope is the most common form of Nitrogen?