Ions in Aqueous Solutions and Colligative Properties
380 likes | 593 Vues
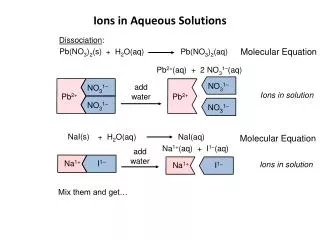
Ions in Aqueous Solutions and Colligative Properties. Chapter 14. Dissociation. The separation of ions that occurs when an ionic compound dissolves. Dissociation examples. You try.

Ions in Aqueous Solutions and Colligative Properties
E N D
Presentation Transcript
Ions in Aqueous Solutions and Colligative Properties Chapter 14 Chemistry chapter 14
Dissociation • The separation of ions that occurs when an ionic compound dissolves. Chemistry chapter 14
Dissociation examples Chemistry chapter 14
You try • Write the equation for the dissolution of NH4NO3 in water. If 1 mol of ammonium nitrate is dissolved, how many moles of each type of ion are produced? • 1 mol of each type of ion Chemistry chapter 14
Precipitation Reactions • When two solutions are mixed, a double replacement reaction may occur. • If one of the products is insoluble, it will form a precipitate. • See table 14-1 on page 427 Chemistry chapter 14
Example • Solutions of (NH4)2S and Cd(NO3)2 are mixed. Will a precipitate form? Chemistry chapter 14
Example continued The cadmium sulfide is the precipitate. Chemistry chapter 14
Net Ionic Equations • Includes only those compounds and ions that undergo a chemical change in a reaction in an aqueous solution. Chemistry chapter 14
Spectator Ions • Ions that do not take part in a chemical reaction and are found in solution both before and after the reaction. Chemistry chapter 14
Example Chemistry chapter 14
You try • A solution of sodium sulfide is combined with a solution of iron(II) nitrate. Does a precipitate form? • Iron(II) sulfide is the precipitate. Chemistry chapter 14
You try continued • Write the net ionic equation for the previous reaction. Chemistry chapter 14
Ionization • The process that forms ions from solute molecules by the action of the solvent. • The attraction between the solvent and the solute is strong enough to break the covalent bonds. Chemistry chapter 14
Hydronium • H3O+ • Formed when an H+ ion is combined with a water molecule (hydrated). • Happens instantly when H+ ions are in water. • Highly exothermic • Formed by many molecular compounds that ionize Chemistry chapter 14
A more accurate picture Chemistry chapter 14
Strong electrolytes • Any compound whose dilute aqueous solutions conduct electricity well. • All or almost all dissolved compound is in the form of ions • Not all compound has to dissolve, but the part that does must be ions Chemistry chapter 14
Weak electrolytes • Any compound whose dilute aqueous solutions conduct electricity poorly. • A small amount of the dissolved compound is in the form of ions. Chemistry chapter 14
Be careful! • Strong electrolytes have a high degree of ionization or dissociation, regardless of their concentration. • Weak electrolytes have a low degree of ionization or dissociation, regardless of their concentration. Chemistry chapter 14
Colligative Properties • Properties of solutions that depend on the concentration of solute particles, but not the identity of solute particles. Chemistry chapter 14
Nonvolatile substance • Has little tendency to become a gas under existing conditions. Chemistry chapter 14
Vapor-pressure lowering • The vapor pressure of a solvent containing a nonvolatile solute is lower than the vapor pressure of the pure solvent at the same temperature. • The solute lowers the concentration of solvent molecules at the surface. • Fewer molecules enter the vapor phase. Chemistry chapter 14
Effects • See figure 14-6 on page 436 • The solution remains liquid over a wider temperature range. • The freezing point is lowered and the boiling point is raised. Chemistry chapter 14
Molal freezing-point constant • Kf • The freezing point depression of the solvent in a 1-molal solution of a nonvolatile, nonelectrolyte solute. • = -1.86 °C/m for water • 2 molal decreases 3.72 °C Chemistry chapter 14
Freezing-point depression • The difference between the freezing points of the pure solvent and a solution of a nonelectrolyte in that solvent. • It is directly proportional to the molal concentration of the solution. Chemistry chapter 14
Example • Determine the freezing point of a water solution of fructose, C6H12O6 made by dissolving 58.0 g of fructose in 185 g of water. • -3.24 °C Chemistry chapter 14
You try • Determine the molal concentration of a solution of ethylene glycol, HOCH2CH2OH, if the solution’s freezing point is -6.40 °C. • 3.44 m Chemistry chapter 14
Molal boiling-point constant • The boiling-point elevation of the solvent in a 1-molal solution of a nonvolatile, nonelectrolyte solute. • Kb = 0.51 °C/m for water Chemistry chapter 14
Boiling-point elevation • The difference between the boiling points of the pure solvent and a nonelectrolyte solution of that solvent. • Directly proportional to the molal concentration of the solution Chemistry chapter 14
Example • What is the boiling point of a solution of 25.0 g of 2-butoxyethanol, HOCH2CH2OC4H9, in 68.7 g of ether? • 40.8 °C Chemistry chapter 14
You try • What mass of glycerol, CH2OHCHOHCH2OH, must be dissolved in 1.00 kg of water in order to have a boiling point of 104.5 °C? • 810 g Chemistry chapter 14
Electrolytes • 1 mole of an electrolyte produces more than one mole of particles in solution. • The ions separate Chemistry chapter 14
Example • A water solution contains 42.9 g of calcium nitrate dissolved in 500. g of water. Calculate the freezing point of the solution. • -2.92 °C Chemistry chapter 14
You try • What is the expected boiling point of a 1.70 m solution of sodium sulfate in water? • 102.6 °C Chemistry chapter 14
Actual values • Our expected values are not always what is observed. • See table 14-3 on page 445 • Differences are caused by attractive forces between ions in solution. Chemistry chapter 14
Semipermeable membrane • Allows the movement of some particles while blocking the movement of others. • Example: allows water molecules through, but not sucrose molecules Chemistry chapter 14
Osmosis • The movement of solvent through a semipermeable membrane from the side of lower solute concentration to the side of higher solute concentration. Chemistry chapter 14
Osmotic pressure • The external pressure that must be applied to stop osmosis. • The greater the concentration of a solution, the greater the osmotic pressure. Chemistry chapter 14