Chemical Reactions
220 likes | 404 Vues
Chemical Reactions. Chemical Reactions. Chemical Reactions happen when atoms join together in a process called Chemical Bonding. Chemical Bonding happens in the outer energy levels of the reacting atoms. Definitions.

Chemical Reactions
E N D
Presentation Transcript
Chemical Reactions Chemical Reactions happen when atoms join together in a process called Chemical Bonding. Chemical Bonding happens in the outer energy levels of the reacting atoms.
Definitions • Compound – two or more elements chemically bonded together. • Molecule – two or more atoms chemically bonded together. (Formed when atoms “share” electrons.) So What is the Difference? A molecule is formed when two or more atoms join together chemically. A compound is a molecule that contains at least two different elements. All compounds are molecules but not all molecules are compounds. Molecules can be two atoms of the SAME element joined together.
More Definitions • Ion – an atom that has gained or lost electrons. Positive IonThe atom has lost electrons giving it a “net” (overall) positive charge. Negative IonThe atom has gainedelectrons giving it a net negative charge.
Types of Chemical Bonds Covalent Bond – the reacting atoms SHARE electrons. (Each atom fills its outer energy level by sharing.) Kindergarten Ionic Bond – the reacting atoms TRANSFER electrons. (Each atom fills its outer energy level by lending or borrowing electrons to fill its outer energy level.) Business Deal
Covalent Bond Ain’t it Cute! They’re Sharing! Kindergarten! Each atom fills its outer energy level by sharing its electrons
Ionic Bond Business Deal! One atom lends – the other atom borrows!
Other Types of Bonds! Metallic Bond – a bond that occurs between metals. (The outer energy levels of the reacting atoms form a common electron cloud.
Coolest Spy? Maxwell Smart and Agent 99 James Bond Austin Powers
Naming Chemical Compounds All compounds must contain at least one metal and one nonmetal. The simplest type of compound is called a BINARY COMPOUND. Just like a Bicycle has two wheels. A Binary Compound is made up of two elements The prefix ‘Bi’ means two!
How to Name a Binary Compound Binary CompoundOne Metal – One Nonmetal • The name of the metal (element with the positive oxidation number) is first: • The name of the element the nonmetal (element with the negative oxidation number) is changed to end with – “ide”. Oxidation number – the number of electrons an atom will transfer or share when forming compounds.
Sodium Chlorine Sodium chloride Metal 1+Don’t touch it! Must be stored in oil to prevent reaction with air or moisture. Nonmetal 1-Breathing in just a little bit can kill you! TABLE SALT
How to use Oxidation Numbers to write Correct Chemical Formulas Atoms and ions always combine in definite ratios. Oxidation numbers can help you to determine these ratios. If the Oxidation numbers of the elements in a compound add up to Zero (0) – then the formula is correct!
Writing Correct Chemical Formulas Na Cl 1+ 1+ 1- 1- One atom of Sodium has one electron to lend One atom of Chlorine needs to borrow one electron 1+ + 1- = 0
Polyatomic Ions Polyatomic ions – a group of atoms that act as one. The prefix ‘poly ‘ means many! A polyatomic ion is the opposite of Sybil. Even though a polyatomic ion is a group of atoms you treat it as if it were only one. Sybil had 16 different personalities. The doctors had to treat her as if she were 16 different people. (One person treated as many people.) Acetate (C2H3O2) is made up of 7 atoms but it has a single oxidation number. 1-
LAW OF CONSERVATION OF MASS(Also known as the Law of Conservation of Matter) Antoine LavoisierFather of Modern Chemistry(1743 – 1794) “In ordinary chemical reactions matter can neither be created nor destroyed.” The number and kind of atoms must be equal for the Reactants and Products in a chemical reaction. The total mass remains the same. “What you start with is what you end up with.” Count the atoms!
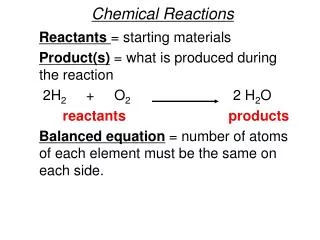
Chemical Equations Reactants - the starting substances in a chemical reaction. (Usually written behind the arrow – Left) 2H2 + O2 2H2O Products - the resulting substances in a chemical reaction. (Usually written in frontof the arrow - Right) Energy is always involved in a chemical reaction. Endothermic – heat energy is taken in (absorbed)Exothermic – heat energy is given off (released – exits )
Chemical Equations are written to show changes that take place in Chemical Reactions. You must “Obey the Law”.The number of atoms of each element must be the same on both sides of the equation. O2 2 H2 + HO 2 2 The plus sign (+) indicates that the reactants have not yet combined. The Arrow means to “yield or produce”. A Subscript shows how many atoms of an element are present in a compound. A Coefficient is a number written in front of symbols or formulas. Coefficients are used to “BALANCE” chemical equations. Don’t forget! You use Oxidation Numbers to write correct chemical formulas.
Five Easy Steps to Balancing Chemical Equations 1. Write the equation in words. Sodium and Chlorine Yields Sodium chloride 2. Write the correct Symbols and Formulas Na + Cl NaCl 1+ 1- Use Oxidation Numbers to write correct chemical formulas. 1+ + 1- = 0
Five Easy Steps to Balancing Chemical Equations 3. Identify any diatomic molecules “Twins” Na + Cl NaCl 2 Many elements do not exist in nature as single atoms. A Diatomic Molecule is made of two atoms of the same element. “Twins” H2 Cl2 O2 I2 Br2 N2 F2
Five Easy Steps to Balancing Chemical Equations 4.Count the Atoms! Na + Cl2 NaCl Two atoms of chlorine One atom of sodium One atom of sodium One atom of chlorine Is the equation balanced? Are the number of atoms of each element the same on both sides of the arrow?
Five Easy Steps to Balancing Chemical Equations 5. “Obey the Law” Use Coefficients to balance the chemical equation Na + Cl2 NaCl 2 2 Sodium - 2 Chlorine - 2 Chlorine - 2 Sodium - 2 Coefficients are written in front of symbols and formulas to balance chemical equations.