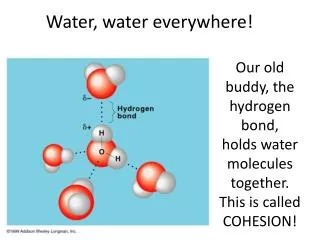
Water
Water. How much water is on earth, and what kind of water is it? Activity 1. The Uses of Water. In your notebook create a list of the ways that people use this precious water? Think hard!

Water
E N D
Presentation Transcript
How much water is on earth, and what kind of water is it? • Activity 1
The Uses of Water • In your notebook create a list of the ways that people use this precious water? Think hard! • After you have at least 10 ways that you use water turn to someone next to you and share and compare your lists. Be sure to add your partners uses to you list and vice versa.
The Uses of Water • Did you include the following in your list: • Water for irrigation(farming). • Water for manufacturing clothing. • Soft drinks contain water, as well as the process of making the bottles they come in. • In mining, water is often used to separate the metal/minerals from the rest of the rock. This metal can be found in the clothing you wear and the electronics that you use.
Water in Irrigation • How can water use for irrigation be helpful and harmful?
Questions to Consider • 1. Which countries are most likely to rely on irrigation to grow crops? Least likely? • 2. In which countries is agriculture probably the main source of commercial income? • 3. What types of domestic, or personal water systems are countries like Cambodia and Gambia likely to have? (hint: Cambodia has 6 litres of water per person per day, Gambia has 3 litres of water per person per day, Canada has…431 litres per person per day!)
Cambodian boy using a local well to get water for his family.
Uses of water • Over 1.4 billion people currently live in river basins where the use of water exceeds minimum recharge levels, leading to the disappearance of rivers and depletion of groundwater.
Facts of Water Use • The world's 6.9 billion people are appropriating 54 percent of all the accessible freshwater contained in rivers, lakes and underground aquifers.
Staggering Statistics • The world’s population is growing by roughly 80 million people each year. • Changes in lifestyles and eating habits in recent years are requiring more water consumption per capita. • The production of biofuels has also increased sharply in recent years, with significant impact on water demand. Between 1,000 and 4,000 litres of water are needed to produce a single litre of biofuel!
Staggering Statistics • Energy demand is also accelerating, with corresponding implications for water demand. • Freshwater withdrawals have tripled over the last 50 years. • Almost 80% of diseases in so called "developing" countries are associated with water, causing some three million early deaths. For example, 5,000 children die every day from diarrhea, or one every 17 seconds.
If you were looking at the earth from the moon you would see mostly blue, a bunch of white and to a lesser extent; greens and brown. The majority of our planet is covered in water, unlike other planets we have water in abundance. • The water we have allowed life to flourish and evolve over millions of years to where we are now. We owe our existence to water.
Water exists naturally in all three states on the earth’s surface. • What are the 3 states of water? • Solid • Liquid • Gas
Water in Liquid State • Nearly all the water on earth is in liquid form for the majority of the time. • Ocean water is home to many varieties of animals and plants but is too salty for many creatures that live on land to drink. • Land animals and plants rely on lakes, rivers, streams and ground water for use.
Water in a Solid State • Most of the fresh water on the planet can be found frozen in glaciers, on mountains, as icebergs, and as snow and ice. • Antarctica and Greenland are two places that contain ice up to 4.8 km. • Many places around the globe ice can be found as a solid for a portion of the year.
Water in a Gaseous State • Only a tiny portion of water can be found as a gas called water vapour. • Often you cannot see the water vapour but you can sometimes feel it as humidity. • On hot summer days that have high humidity you will feel damp and clammy. • Clouds form when water vapour condenses to form tiny droplets of water or ice crystals.
Questions This picture was taken near St. John’s, Newfoundland during the summer months. Where do you think the iceberg came from? How do you think it was formed? NL Iceberg Action
Salt Water vs. Fresh Water Lab • What did we learn about the characteristics of salt water compared to that of fresh water?
Although both sea water and “fresh” water have levels of salt it is clear that sea water is much more salty. The average salt content in sea water is about 3.5%. In addition to salt, sea water has many other substances in smaller amounts. Even gold and silver!
What other substances can be found in fresh and salt water? • In parts of Ontario, water contains so much iron that it affects the taste. • Too much calcium and magnesium makes water “hard” so that it is difficult to lather with soap. • GNB Media Release
Did you know? The places where fresh water from rivers flows into salt water oceans are called brackish water. The salt content is lower then the ocean but higher then the river. Only certain organisms can live in these water ways.
What does salt do to water? • Have you noticed that the ocean doesn’t really freeze, unlike lakes and ponds that tend to freeze more often? • One reason why this happens in because salt in water(salinity) changes its freezing point. • This is why we often put salt on the roads during winter months. • High salinity of water lowers its freezing point. This means salt water needs colder temperatures in order to freeze.
Salt Water Salt is sodium chloride (NaCl) • We’ve talked about the percentage of salt water vs. fresh water found on our planet. (roughly 98% vs. 2%) • We discovered that the salinity of water affects density and buoyancy. • Salinityis the saltiness of a body of water or soil.
Density can be described as the “crowdedness” of the particles that make up matter. In this case, water.
Density • In scientific terms, density is the amount of substance that occupies a particular space. • When you refer to a substance as being light or heavy, you are referring to the property of density.
Density • According to the particle theory, different substances have different sized particles. • The size of the particles determines how many particles can fit into a given space. • Because of this, each substance has its own unique density based on particle size.
Think about this… • Imagine two identical containers, one filled with a liquid(water) and one filled with a gas (water vapour). • Both are the same substance therefore they have particles of the same size. • According to the particle theory, gas particles have more space between them than liquid particles. • It would be reasonable to conclude that the density of water vapor is less than the density of liquid water.
Waves • We have discussed some ways that water can move when we discussed the water cycle, however, water can move other ways too. • Waves, tides, and currents are powerful forces that interact with the environment. • What types of “waves” have you heard of?
Waves • Microwaves, radio waves, sound waves, water waves … • Do these different waves move the same way?
Making Waves Lab – Part 1 • Fill a deep pan with water. • Wait until the surface is calm the blow across the surface to create waves. • Sketch a picture of the waves you’ve created. • Experiment with creating differences in the waves.
Making Waves Lab – Part 2 • Place a drop of food colouring in a shallow pan of water. • Blow across the surface of the water. • Observe how the waves distribute the food colouring around the water. Are the currents you create similar to the current found in the world’s oceans?
Making Waves Lab – Part 3 • Place a cork in a deep pan filled with water. • Wait until the water is calm. • Blow on the water but not on the cork directly. • Can you move the cork across the pan with the waves you’ve created? Why or why not?
Waves • Waves are patterns that move along the surface of the water’s surface. • TIL – The water itself doesn’t move far, it just moves up and down, but the waves can travel many kilometres across the surface of the ocean! • Skipping rope demonstration
Solar Powered Waves • Waves, believe it or not, start from the sun! • The sun doesn't ever heat the Earth evenly. Some spots get heated more than others. • As some air gets heated, it becomes less dense, and thus lighter, and naturally floats upward. This leaves an open space for denser, colder air to rush in and take its place. • This air rush is the refreshing cool breeze you feel on a sunny day.
Wind and Waves • Wind is also responsible for our very powerful waves. As wind rushes up along the water, the friction causes ripples. • Wind continues to push against these ripples in a snowball effect that eventually creates a large wave. • Essentially, this action is a transfer of energy from the sun to the wind to the waves.
Wave Strength • A few factors determine how strong an individual wave will be. These include: • Speed of wind: The faster the wind is traveling, the bigger a wave will be. • Time of wind: The wave will get larger the longer the length of time the wind is hitting it. • Distance of wind: The farther the wind travels against the wave (known as fetch), the bigger it will be.
Wave Parts • When waves make their way across the water surface, the wave swells larger. The top of the wave is called the crest. • The bottom of the wave is called the trough. • The distance between them is called the wave height and the space between each crest is called the wavelength.
Waves and Energy • When waves make their way across water, it is the energy moving, not the water! • If you were in the ocean on a day with many waves, you wouldn’t move any great distance, you would just bob up and down in a circular motion!
Questions • Why are rocks and stones found on beaches often rounded and smooth? • Would it take a long time or a short time for a note in a bottle to travel to land if thrown from a ship at sea? Explain. • Why are waves a problem for people who live close to the shoreline?