Carboxylic Acid Derivatives
E N D
Presentation Transcript
1. Carboxylic Acid Derivatives Bettelheim, Brown, Campbell and Farrell
Chapter 19
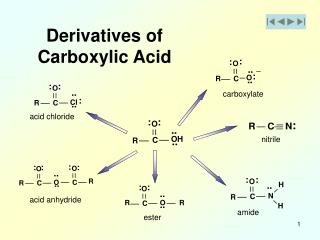
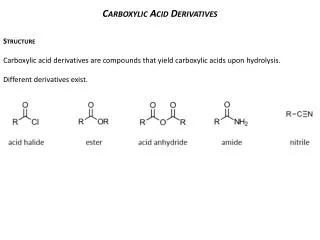
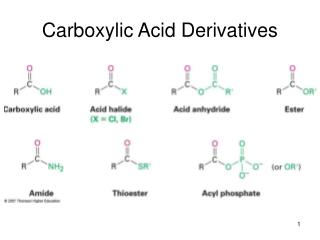
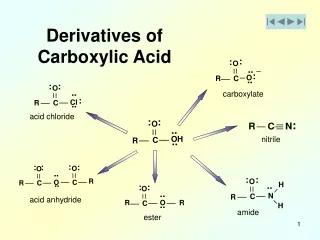
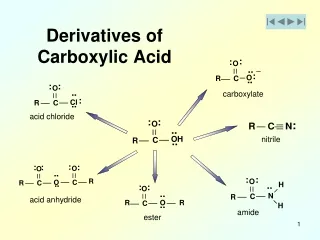
2. Derivatives of Carboxylic Acids Carboxylic acids
Derivatives of carboxylic acids
Anhydrides, Esters, and Amides
Made by reacting a carboxyl acid group with another molecule. H2O is formed in each reaction
3. Properties of Organic Families: ACIDIC BASIC NEUTRAL
carboxylic amines alcohols
acids carboxylate ethers
phenols ions aldehydes
thiols ketones
amides
esters
4. Esters An ester has a carbonyl group bonded to an -OR group
Ester names are derived from the names of the parent carboxylic acids
Name the alkyl or aryl group bonded to oxygen first, followed by the name of the acid; replace the suffix -ic acid by -ate
Cyclic ester is called a lactone
6. Making an ester Esterification
Needs acid catalyst (i.e., conc H2SO4)
Reversible reaction
7. Fischer Esterification Fischer esterification is commonly used to make esters
Carboxylic acid is reacted with an alcohol in the presence of an acid catalyst, such as concentrated sulfuric acid
Fischer esterification is reversible
Can drive reaction in either direction by altering experimental conditions (Le Chatelier�s principle)
8. Fischer Esterification Alcohol adds to the carbonyl group of the carboxylic acid to form a tetrahedral carbonyl addition intermediate
Intermediate then loses H2O to form an ester
9.
10. Hydrolysis of Esters Break ester into carboxylic acid and alcohol
Add HOH to break bond
Reverse of esterification
Acid Hydrolysis: Carboxylic acid + alcohol
Basic Hydrolysis: Carboxylate ion + alcohol Saponification
11. Hydrolysis of Esters Occurs very slowly in water
Much more rapid when ester is heated in aqueous acid or base
Acidic hydrolysis of esters is the reverse of Fischer esterification
Large excess of water drives the equilibrium to the right to form the carboxylic acid and alcohol (Le Chatelier's principle)
12. Hydrolysis of Esters Basic hydrolysis of ester called saponification
Used in the manufacture of soaps
Basic hydrolysis done in hot aqueous base, such as aqueous NaOH
Ester hydrolysis forms carboxylic acid and alcohol
Carboxylic acid reacts with hydroxide ion to form a carboxylate ion
One mole of base needed for each mole of ester
14. Phosphoric Esters Phosphoric acid forms mono-, di-, and triphosphoric esters
In more complex phosphoric esters, it is common to name the organic molecule and then indicate the presence of the phosphoric ester by either the word "phosphate" or the prefix phospho-
Dihydroxyacetone phosphate and pyridoxal phosphate are shown as they are ionized at pH 7.4, the pH of blood plasma
15. Amides An amide has a carbonyl group bonded to a nitrogen atom
Amide names:
IUPAC: Drop the suffix -oic acid and add -amide
Common: Drop the suffix -ic acid and add -amide
Alkyl or aryl groups attached to the N show their location with an N- followed by the name of group
Two alkyl or aryl groups are shown by N,N-di-
18. Amides Cyclic amide is called a lactam
Penicillins are referred to as b-lactam antibiotics
19. Amides the cephalosporins are also b-lactam antibiotics
20. Preparation of Amides Theoretically can form of an amide by reacting a carboxylic acid with an amine
Remove -OH from the acid and an -H from the amine
In practice, acid and amine undergo acid-base reaction to form ammonium salt.
Can then heat salt to high temperature to form amide (water is eliminated)
21. Making an ester Esterification
Needs acid catalyst (i.e., conc H2SO4)
Reversible reaction
22. Making an Amide Generally prepare amides by reacting an amine with an anhydride
23. Hydrolysis of Amides Amides require more vigorous conditions for hydrolysis in both acid and base than do esters
Hydrolysis in hot aqueous acid gives a carboxylic acid and an ammonium ion
Hydrolysis is driven to completion by the acid-base reaction between ammonia or the amine and the acid to form an ammonium ion
Each mole of amide requires one mole of acid
24. Hydrolysis of Amides Hydrolysis of an amide in aqueous base gives a carboxylic acid salt and ammonia or an amine
Hydrolysis is driven to completion by the acid-base reaction between the carboxylic acid and base to form a salt
Each mole of amide requires one mole of base
25. Anhydrides An anhydride has two carbonyl groups bonded to the same oxygen
Symmetrical: two identical acids OR
Mixed: two different acids
Naming: Drop the word "acid" from the name of the carboxylic acid and add the word "anhydride"
26. Formation of Anhydride
27. Hydrolysis of Anhydrides Hydrolysis: break bond by adding water
Carboxylic anhydrides react readily with water to give two carboxylic acids
Low-molecular-weight anhydrides react very easily
28. Phosphoric Anhydrides A phosphoric anhydride has two phosphoryl (P=O) groups bonded to the same oxygen atom
29. Phosphoric Anhydrides Energy storage (ATP)
30. Polymers Addition polymers
Have C=C double bonds
Add each monomer to end of growing chain
Step-Growth polymers
Also called condensation polymers
Small molecule removed when bond between monomers forms: H2O, HCl, etc.
Formed by reaction of two different functional groups on monomers
32. Types of Step-Growth Polymers Found in Nature Polysaccharides:
Individual sugars joined by glycosidic bonds
Proteins:
Amino acids joined by amide bonds
Nucleic Acids:
Nucleotides joined by phosphodiester bonds
33. Step-Growth Polymers Step-growth polymers are formed by reaction between molecules containing two functional groups, with each new bond created in a separate step
Different types of step-growth polymers
Polyamides
Polyesters
Polycarbonates
34. Polyamides Nylon-66 was the first purely synthetic fiber
Synthesized from two six-carbon monomers
35. Polyamides Kevlar is made from an aromatic dicarboxylic acid and an aromatic diamine
36. Polyesters First polyester involved polymerization of this diacid and ethylene glycol
37. Polycarbonates Lexan, the most familiar polycarbonate, is formed by reaction between the disodium salt of bisphenol A and phosgene