States of Matter: Chapter Review and Test Prep
100 likes | 127 Vues
This session will go over answers to the States of Matter worksheet and key concepts from the textbook. Prepare for the test covering scientific method, metric conversion, and more. Topics include mixtures, pure substances, and composition of matter. Get ready for Friday's test with our review materials.

States of Matter: Chapter Review and Test Prep
E N D
Presentation Transcript
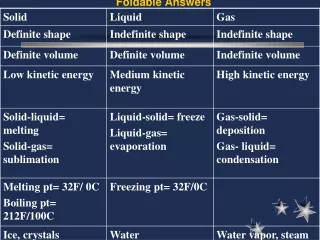
Finish States of Matter WS: will go over answers together • Textbook: • 465 1-4,p. 483 1-3 • Read chapter 15 and use notes: p. 456 1-2, p. 465 1-3, p. 470 1-10, 14-16, • Pick up Basic of Science WS • Pick up Review Sheet • States of Matter WS • Test Friday: scientific method, metric conversion, density, states of matter, properties
MIXTURE PURE SUBSTANCE yes no yes no Is the composition uniform? Can it be decomposed by chemical means? A. Matter Flowchart MATTER yes no Can it be separated by physical means? Homogeneous Mixture (solution) Heterogeneous Mixture Compound Element
MATTER Ch. 9 - Classification of Matter II. Composition of Matter (p.246-250) Matter Flowchart Pure Substances Mixtures
B. Pure Substances • Element • matter composed of identical atoms • EX: copper
B. Pure Substances • Compound • matter composed of 2 or more elements in a fixed ratio • properties differ from those of individual elements • EX: salt (NaCl)
C. Mixtures • Variable combination of 2 or more pure substances. • Homogeneous Mixture (Solution) • even distribution of components • very small particles • particles never settle • EX: saline solution
C. Mixtures • Heterogeneous Mixture • uneven distribution of components • colloids and suspensions • EX: granite
C. Mixtures • Colloid • medium-sized particles • Tyndall effect - particles scatter light (looks cloudy) • particles never settle • EX: milk
C. Mixtures • Suspension • large particles • particles scatter light • particles will settle (needs to be shaken) • EX: fresh-squeezed lemonade