Fibrinogen
700 likes | 812 Vues
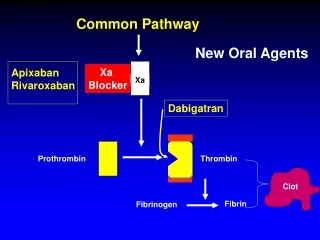
Xa. Common Pathway. New Oral Agents. Xa Blocker. Apixaban Rivaroxaban. Dabigatran. Prothrombin. Thrombin. Clot. Fibrin. Fibrinogen. Replacing Traditional Anticoagulants. Treatment VTE UFH, LMWH Bridge to Warfarin. RE-COVER Study. Dabigatran 150 mg, BID for 6 months

Fibrinogen
E N D
Presentation Transcript
Xa Common Pathway New Oral Agents Xa Blocker Apixaban Rivaroxaban Dabigatran Prothrombin Thrombin Clot Fibrin Fibrinogen
RE-COVER Study Dabigatran 150 mg, BID for 6 months Double Blind, Double Dummy, Non-Inferiority Schulman S, et al NEJM 2009;361:2342-2352
RE-COVER Study VTE Major Bld 2.4% 1.6% Dabigatran 150 mg, BID VTE 2.1% 1.9% Warfarin INR 2-3 6 months Parenteral Anticoagulant Median 9 days Warfarin TTR= 60% Schulman S, et al NEJM 2009;361:2342-2352
RE-COVER StudyIndex Events Dabi 1273 Warfarin 1266 Schulman S, et al NEJM 2009;361:2342-2352
RE-COVER StudyMajor Bleeding Warfarin Dabi Schulman S, et al NEJM 2009;361:2342-2352
RE-COVER • A limitation of the study is that the first dose of dabigatran, was given only after initial parenteral anticoagulation therapy had been administered for median of 9 days • “There is no data to support the use of dabigatran monotherapy for acute venous thromboembolism” Schulman S, et al NEJM 2009;361:2342-2352
Rivaroxaban 15 mg, PO, BID x 3 weeks then 20mg, Qday Enoxaparin 1mg/kg/Q12hrs bridge to Warfarin INR 2-3 Open Label, Non-Inferiority trial Einstein Investigators NEJM 2010;363:2499-2510
Einstein DVT Rivaroxaban 15 mg, BID x 3 wks 20 mg, Qday VTE Major Bld 2.1% 8.1% DVT Enoxaparin Warfarin INR 2-3 3.0% 8.1% 3, 6, 12 months Proximal DVT Warfarin TTR = 57.7% Einstein Investigators NEJM 2010;363:2499-2510
Einstein Acute DVT StudyCauses of VTE Riva Standard Einstein Investigators NEJM 2010;363:2499-2510
Einstein Acute DVT StudySafety Outcomes Riva Standard Einstein Investigators NEJM 2010;363:2499-2510
Rivaroxaban 15 mg, PO, BID x 3 weeks then 20mg, Qday Enoxaparin 1mg/kg/Q12hrs bridge to Warfarin INR 2-3 Open Label, Non-Inferiority Einstein Investigators NEJM 2012;366:1287-1297
Einstein PE Rivaroxaban 15 mg, BID x 3 wks 20 mg, Qday VTE Major Bld 2.1% 1.1% PE Non-Inferior Enoxaparin Warfarin INR 2-3 1.8% 2.2% 3, 6, 12 months Warfarin TTR = 62.7% Einstein-PE Investigators NEJM 2012;366:1287-1297
Einstein PECauses Riva Standard Einstein Investigators NEJM 2012;366:1287-1297
Einstein PEAnatomical Extent Standard Riva Einstein Investigators NEJM 2012;366:1287-1297
ED - OBS History & Physical Laboratory Testing Diagnosis DVT Select Treatment Hospital Admission OBS Discharge Plan Secure Rx Communication Follow Up Acquire Med Contact PCP Phone call 24 hrs Pt Education D/C Summary Appointment 3-5 days Discharge OBS
Your patient who has been on long term warfarin would like to convert to one of the new oral anticoagulant.
Rivaroxaban 15 mg, PO, BID x 3 weeks then 20mg, Qday Enoxaparin 1mg/kg/Q12hrs bridge to Warfarin INR 2-3 Open Label, Non-Inferiority trial Einstein Investigators NEJM 2010;363:2499-2510
Einstein DVT-Extend Rivaroxaban 20 mg, Qday VTE Major Bld 1.3% 0.7% All Rxed DVT Placebo 7.1% 0% 3, 6, 12 mo 6-12 mo Einstein Investigators NEJM 2010;363:2499-2510
Double Blind, Randomized Trial Schulman S, et al NEJM 2013;368:709-718
RE-MEDY VTE Major Bld Dabigatran 150 mg, BID 1.8% 0.9% VTE Warfarin INR 2-3 1.3% 1.8% 6 months Patient Rx 3 to 12 months Schulman S, et al NEJM 2013;368:709-718
RE-SONATE VTE Major Bld Dabigatran 150 mg, BID 0.4% 0.3% DVT Placebo 5.6% 0% 6-18 months Patient Rx 6 to 18 months Schulman S, et al NEJM 2013;368:709-718
RE-SONATE Study Schulman S, et al NEJM 2013;368:709-718
AMPLIFY-EXT VTE Major Bld Apixaban 2.5 mg, BID 1.7% 0.2% 1.7% 0.1% Apixaban 5.0 mg, BID VTE 8.8% 0.5% Placebo Rx 6-12 mo 12 months Agnelli G, et al NEJM 2013;368(8):699-708
AMPLIFY-EXT Apixaban 5 Placebo Apixaban 2.5 Agnelli G, et al NEJM 2013;368(8):699-708
Warfarin to NOAC NOAC= New Oral Anticoagulants
Atrial Fibrillation Studies Time Therapeutic Range = TTR Modified Ahrens I, et al Thromb Haemost 2011;105
Atrial Fibrillation StudiesWhen should new orals be started? • RE-LY (Dabigatran) • Stroke within 14 days • Severe stroke within last 6 months • ARISTOTLE (Apixaban) • Stroke within 7 days • ROCKET-AF (Rivaroxaban) • Stroke within 14 days • Severe stroke within last 3 months Modified-Ahrens I, et al Thromb Haemost 2011;105
EXCLAIMExtended VTE Px Medically-ill Hull R, et al, Ann Intern Med 2010;153:8-18
ADOPT Apixaban 2.5 mg BID Enoxaparin 40mg, Qday Goldhaber S, et al NEJM 2011;365(23):2167-2177
ADOPT Study Goldhaber S, et al, NEJM, 2011; 365: 2167-2177
MAGELLAN Study Cohen A, et al NEJM 2013;368:513-523
Extended VTE Prophylaxis In Medical Patients Net Clinical Benefit of Factor Xa Inhibitors EXCLAIM ADOPT MAGELLAN * p < 0.05 6 * p < 0.05 * 0.3 0.8* 3 1.7 2.1 * 2.7 Incidence (%) 0 4.1* 3 (Major Bleeding) 6 (n = 5,963) (n = 6,528) (n = 8,101) Hull R, et al, Ann Intern Med 2010;153:8-18 Cohen A, et al NEJM 2013;368:513-523 Goldhaber S, et al, NEJM, 2011; 365: 2167-2177
Key PointsBlack Box Warnings, Stroke, MI Risk, Drug Interactions, Major Bleeding
ROCKET AF Patel M et al, NEJM 2011;365:883-891
Rocket AF Study Patel M, et al JACC 2013;61:651-658
Interruption or DiscontinuationRivaroxaban • Temporary Interruption (Events starting 3 days after interruption until 3 days after resumption) • Early Permanent Study Drug Discontinuation (Events evaluated from 3-30 days after d/c) • End of Study Transition to Open-Label (Events evaluated from 3-30 days after d/c)
Rocket AF Study Warfarin 81% 49% Rivaroxaban Cumulative Proportion with INR > 2 Days after Last Dose at End of Study Patel M, et al JACC 2013;61:651-658