Modeling Surface Phase Activity Coefficients and Reaction Equilibria in Aqueous Systems
60 likes | 199 Vues
This study presents a detailed analysis of surface phase activity coefficients in aqueous systems through logarithmic equations for equilibrium constants K1 and K2. Using the constant capacitance model, we derive expressions for logK in terms of activity coefficients and electrochemical potential. Key relationships reveal how pH influences the equilibrium constant K2 under PZNPC conditions, establishing that K2 is inversely proportional to (H+)². This work emphasizes the role of surface phenomena in chemical reactivity, providing insights valuable for researchers in surface chemistry and electrochemistry.

Modeling Surface Phase Activity Coefficients and Reaction Equilibria in Aqueous Systems
E N D
Presentation Transcript
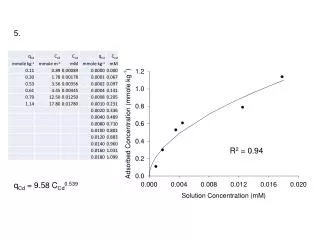
5. R2 = 0.94 qCd = 9.58 CCd0.539
ln qCd = 3.02 + 0.695 ln CCd qCd = 20.49 CCd0.695 qCd = 9.58 CCd0.539 R2 = 0.90
12. σH = qH – qOH = [SOH2+] – [SO-] – [(SO)2Ca0] σIS = 2[(SO)2Ca0] – [SHPO4-] σOS = 0 σD = -[SOH2+] + [SO-] + [SHPO4-]
If SO- + H+ = SOH K1 SO- + 2H+ = SOH2+ K2 Develop log equations for K in terms of log cK and constant capacitance model surface phase activity coefficients. K1 = {[SOH] / [SO-] (H+)} exp (ΔzFΨ0 /RT) where Δz = 0 – (-1) = 1 logK1 = logcK + 2.303 (FΨ0 /RT) logcK + 2.303 (F2σP /SCRT) K2 = [SOH2+] / [SO-] (H+)2} exp (ΔzFΨ0 /RT) where Δz = 1 – (-1) = 2 logK2 = logcK + 2.303 (2FΨ0 /RT) logK2 = logcK + 2.303 (2F2σP /SCRT)
Show that PZNPC = ½ logK2 K2 = [SOH2+] / [SO-] (H+)2} exp (ΔzFΨ0 /RT) At PZNPC, [SOH2+] = [SO-] and Ψ0 = 0 • K2 = 1 / (H+)2 So, log K2 = 2pH or PZNPC = ½ log K2