Activated complex theory
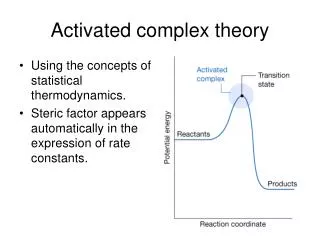
Activated complex theory. Using the concepts of statistical thermodynamics. Steric factor appears automatically in the expression of rate constants. 27.4 The Eyring equation.


Activated complex theory
E N D
Presentation Transcript
Activated complex theory • Using the concepts of statistical thermodynamics. • Steric factor appears automatically in the expression of rate constants.
27.4 The Eyring equation • The transition state theory pictures a reaction between A and B as proceeding through the formation of an activated complex in a pre-equilibrium: A + B -> C‡ K‡ = ( `‡` is represented by `±` in the math style) • The partial pressure and the molar concentration has the following relationship: pJ = RT[J] • thus [C‡] = K‡ [A][B] • The activated complex falls apart by unimolecular decay into products, P, C‡ → P v = k‡[C‡] • So v = k‡ K‡ [A][B] Define k2 = k‡ K‡ v = k2[A][B]
(a) The rate of decay of the activated complex k‡ = κv where κ is the transmission coefficient. κ is assumed to be about 1 in the absence of information to the contrary. v is the frequency of the vibration-like motion along the reaction-coordinate.
(b) The concentration of the activated complex Based on Equation 20.54, we have with ∆E0 = E0(C‡) - E0(A) - E0(B) are the standard molar partition functions. provided hv/kT << 1, the above partition function can be simplified to Therefore we can write qC‡ ≈ where denotes the partition function for all the other modes of the complex. K‡ =
(c) The rate constant combine all the parts together, one gets k2 = k‡ K‡ = κv then we get k2 = κ (Eyring equation)
(d) The collisions of structureless particles A + B → AB Because A and B are structureless atoms, the only contribution to their partition functions are the translational terms: k2 = κ k2 = κ NA
Kinetics Salt Effect Ionic reaction A + B → C‡ C‡→ P d[P]/dt = k‡[C‡] the thermodynamic equilibrium constant Then d[P]/dt = k2[A][B] Assuming is the rate constant when the activity coefficients are 1 ( ) Debye-Huckle limiting law with A = 0.509 log(k2) = log( ) + 2AZAZBI1/2 (Analyze this equation)
Example: The rate constant for the base hydrolysis of [CoBr(NH3)5]2+ varies with ionic strength as tabulated below. What cab be deduced about the charge of the activated complex in the rate-determining stage? I 0.0050 0.0100 0.0150 0.0200 0.0250 0.0300 k/ko 0.718 0.631 0.562 0.515 0.475 0.447 Solution: I1/2 0.071 0.100 0.122 0.141 0.158 0.173 Log(k/ko) -0.14 -0.20 -0.25 -0.29 -0.32 -0.35
27.6 Reaction Collisions • Properties of incoming molecules can be controlled: 1. Translational energy. 2. Vibration energy. 3. Different orientations. • The detection of product molecules: 1. Angular distribution of products. 2. Energy distribution in the product.
27.7 Potential energy surface • Can be constructed from experimental measurements or from Molecular Orbital calculations, semi-empirical methods,……
27.8 Results from experiments and calculations (a) The direction of the attack and separation