Reactions
E N D
Presentation Transcript
Reactions Making sense of chemical equations
Types of Reactions • There are millions of reactions, and we cannot remember them all. Luckily they fall into several categories. • By looking at the reactants, we will: • learn the 5 major types • predict the products • predict if they happen at all
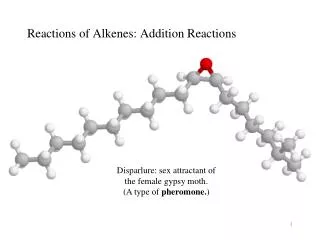
Type 1: Combination • Combination reactions happen when 2 elements combine to make a compound (also called synthesis). • Examples: • Ca + O2 CaO • SO3 + H2O H2SO4
Type 2: Decomposition • Decomposition reactions occur when a compound breaks apart into two or more elements or compounds. • Examples: • NaCl Na + Cl2 • CaCO3CaO + CO2 • **Note that energy is usually required.
Type 3: Single Replacement • In a single replacement reaction, one element replaces another. • Reactants must be one element and one compound. • Products will be a different element and a different compound.
Type 3: Single Replacement • Examples: • Na + KCl K + NaCl • cation replaces cation • F2 + LiCl LiF + Cl2 • anion replaces anion
Type 4: Double Replacement • Double replacement reactions occur when two elements replace each other in two different compounds. • Reactants must be ionic compounds in aqueous solution. • NaOH + FeCl3 Fe(OH)3+ NaCl • The positive ions change places.
How to recognize each type: • Look at the reactants: E + E = Combination C = Decomposition E + C = Single Replacement C + C = Double Replacement E = element; C = compound
Type 5: Combustion • Combustion means “add oxygen”. • Combustion reacts occur when compounds containing C, H, and O react with oxygen – usually called “burning”. • If the combustion reaction is complete, the products will be CO2 and H2O. • If the combustion reaction is incomplete, the products will be CO (possibly just C) and H2O.
Type 1: Combination • We can predict the products, especially if the reactants are two elements: • Mg + N2 • Al + Cl2 Mg3N2 AlCl3
Type 1: Combination • Practice: • Ca + Cl2 • Fe + O2 (assume Fe (II) in the product) • Al + O2
Type 1: Combination • Watch out for: • Some nonmetal oxides react with water to form acids: • SO2 + H2O H2SO4 • **This is what happens to make “acid rain” • Some metallic oxides react with water to for bases: • CaO + H2O Ca(OH)2 • **The hydroxide polyatomic ion indicates that the compound is a base.
Type 2: Decomposition • We can predict the products if it is binary compound (made up of two elements) – it breaks apart into the elements. • Examples: • H2O • HgO
Type 2: Decomposition • If the compound has more than two elements, you must be given at least one of the products. The other product will be made from the missing pieces. • Examples: • NiCO3 CO2 + _______ • H2CO3(aq) CO2 + _______
Type 3: Single Replacement • Metals will also replace other metals (and also hydrogen) • Examples: • K + AlN • Zn + HCl • Think of water at HOH: • Metals replace the first H and then combine with OH-. • Na + HOH
Type 3: Single Replacement • We can even tell whether or not a single replacement reaction will happen: • Because some metals are more “active” than others. • More active replaces less active. • There is a list on page 217 in the textbook. • Called the Activity Series of Metals. • Higher on the list replaces lower on the list.
Type 3: Single Replacement Higheractivity • Lithium • Potassium • Calcium • Sodium • Magnesium • Aluminum • Zinc • Chromium • Iron • Nickel • Lead • Hydrogen • Bismuth • Copper • Mercury • Silver • Platinum • Gold • Rules: • 1) Metals can replace other metals provided they are above the metal they are trying to replace. For example, zinc will replace lead. • 2) Metals above hydrogen will replace hydrogen in acids. • 3) Metals from sodium upward can replace hydrogen in water (HOH). Lower activity
Type 3: Single Replacement • Practice: • Fe + CuSO4 • Pb + KCl • Al + HCl
Type 3: Single Replacement Higher Activity Fluorine Chlorine Bromine Iodine • Halogens have a similar activity series. • Halogens can replace other halogens in compounds if they are above the halogen they are trying to replace. • Examples: • 2NaCl + F2 • MgCl2 + Br2 Lower Activity
Type 4: Double Replacement • Double replacement reactions occur because of certain driving forces (or reasons). The reaction will only happen if one of the products: • doesn’t dissolve in water and forms a solid (a precipitate), or • is a gas that bubbles out, or • is a molecular compound (which will usually be water).
Complete and Balance • assume all of the reactions take place • CaCl2 + NaOH • CuCl2 + K2S • KOH + Fe(NO3)3 • (NH4)2SO4 + BaF2
How to recognize each type: • Look at the reactants: E + E = Combination C = Decomposition E + C = Single Replacement C + C = Double Replacement E = element; C = compound
Practice • H2 + O2 • H2O • Zn + H2SO4 • HgO • KBr + Cl2 • AgNO3 + NaCl • Mg(OH)2 + H2SO3
Type 5: Combustion • C4H10 + O2(assume complete) • C4H10 + O2(incomplete) • C6H12O6 + O2(complete) • C8H8 + O2(incomplete)
SUMMARY • Reactions come in 5 types. • We can tell what type of reaction by looking at the reactants. • Single replacement reactions occur based on activity series. • Double replacement reactions occur if one product is: 1) a precipitate, 2) a gas, or 3) water (a molecular compound).