Calculating Percentage Composition of Compounds
Learn how to determine the percentage composition of compounds by calculating molar mass and dividing element masses, with examples for water, sucrose, and sulfuric acid.

Calculating Percentage Composition of Compounds
E N D
Presentation Transcript
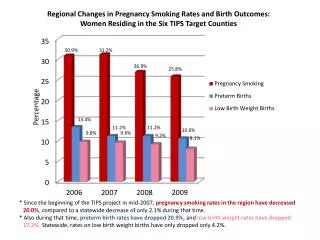
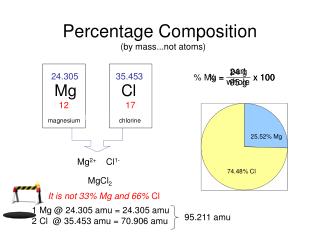
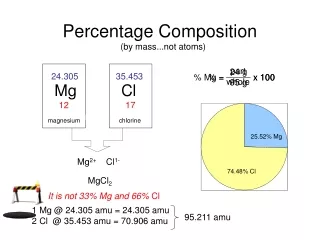
Percentage composition • Indicates the relative amount of each element present in a compound.
Calculating percentage composition • Step 1 : Calculate molar mass • Step 2 : Divide the subtotal for each element’s mass by the molar mass. • Step 3: Multiply by 100 to convert to a percentage.
Example 1 • 1. Calculate the molar mass of water, H2O. • Step 1 – H- 2(1.01)= 2.02 • O – 1(16.0)= 16.0 • ________ • 18.02
Step 2 2.02/18.02 X 100 = 11.21% • 16.0 /18.02 X 100 = 88.79 % • Water is composed of 11.21 % hydrogen and 88.79 % oxygen.
Example 2 • Calculate the percentage composition of sucrose , C12H22O11.
Example 3 • Find the percentage composition of hydrogen in sulfuric acid.