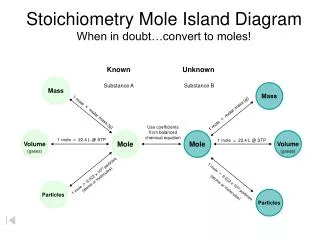
Molar Mass & Stoichiometry
This guide provides an overview of molar mass calculations and stoichiometry involving various compounds. Key examples include determining the molar mass of common compounds such as Mg(OH)2 and H3PO4, converting mass to moles for sulfur and LiBr, and deriving stoichiometric yield in reactions like PbS with O2 and Al with O2. It also covers percent composition and empirical formulas for compounds, including determining the empirical formula based on mass percentages. This comprehensive resource is designed for chemistry students looking to enhance their understanding of these fundamental concepts.

Molar Mass & Stoichiometry
E N D
Presentation Transcript
What is the Molar Mass of Mg(OH)2 ? • 41.32 g/mole • 42.33 g/mole • 58.33 g/mole • 827.03 g/mole • Not listed
What is the Molar Mass of H3PO4? • 47.98 g/mole • 95.96 g/mole • 98.00 g/mole • 196.00 g/mole • Not listed
Convert 125.25 g of Sulfur (S8) (MM = 256.56 g) to moles. • 0.48819 moles • 3.9055 moles • 4.4588 moles • 157.32 moles • 4016.8 moles • Not listed
Convert 3.75 moles of BaSO4 to grams.(MM = 233.4 g) • 875 g • 233.40 g • 62.2 g • 0.0161 g • Not listed
Convert 2.50 moles of KCl (MM = 74.55 g) to molecules. • 4.15 x 10-24molecules • 1.51 x 1024 molecules • 6.02 x 1023 molecules • 6.78 x 1025 molecules • Not listed
Convert 70.0 g of LiBr (MM = 86.84 g) to molecules. • 7.47 x 1023 molecules • 4.21 x 1025 molecules • 3.65 x 1027molecules • 4.85 x 1023molecules • Not listed
Given the reaction2 PbS + 3 O2 2 PbO + 2 SO2239.27 32.00 223.20 64.07 g/mole • 261.6 grams • 116.3 grams • 174.4 grams • 223.2 grams • 186.9 grams • Not listed 25.00 g of O2 will yield how many grams of PbO?
Given the reaction4 Al + 3 O2 2 Al2O326.98 32.00 101.96 g/mole • 920 grams • 6200 grams • 460 grams • 61 grams • 230 grams • Not listed 4.5 moles of Al will yield how many grams of Al2O3?
Given the reaction2 C6H14 + 19 O2 12 CO2 + 14 H2O86.20 32.00 44.01 18.02 g/mole • 5.9354 x 1026 molecules • 8.4792 x 1025 molecules • 1.2113 x 1025 molecules • 6.8856 x 1024 molecules • 2.8812 x 10-21 molecules • Not listed 140.85 grams of C6H14 will yield how many molecules of H2O?
What is the % mass of lead in PbO2? • 86.62% • 92.83% • 13.37% • 6.69% • 13.37% • Not listed
What is the % mass of Cu in Cu2S? • 66.46% • 39.93% • 79.85% • 20.14% • 33.54% • Not listed
What is the % mass of nitrogen in Ba(NO3)2? • 5.36% • 10.72% • 16.08% • 47.45% • 23.73% • Not listed
What is the mass of oxygen in 54 g of H2O. • 48 g • 16 g • 6.0 g • 3.0 g • Not listed
A compound is found to contain 53.70% iron and 46.30% sulfur. What is the empirical formula of this compound? • FeS • FeS2 • Fe2S3 • Fe3S2 • Not listed
A compound contains 4.04 g of N and 11.46 g of O. What is the compound’s empirical formula? • NO • N2O7 • N5 O2 • N2O5 • Not listed
The molar mass of a compound is 283.89 g/mol. If the empirical formula is P2 O5, what is its molecular formula? • P4 O10 • P4 O5 • P10O50 • P2O5 • Not listed