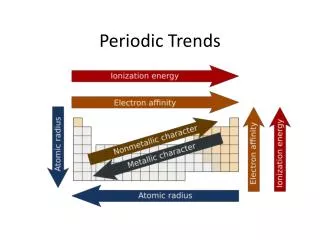
Periodic Trends
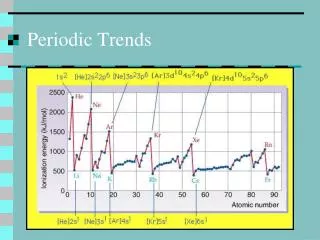
This guide explores periodic trends using the Bohr model of the atom. Noble gases exhibit a complete outer electron shell, indicating stability. Families in the periodic table share the same number of valence electrons, leading to similar chemical properties. As you move across the periodic table, the number of valence electrons and protons increases, influencing element behavior. Additionally, the number of energy levels corresponds to the period of an element, with Period 1 having one energy level and Period 2 having two. Delve into how these factors shape the periodic table's organization.

Periodic Trends
E N D
Presentation Transcript
Periodic Trends Using the Bohr model to find Periodic Trends
The Noble Gases have a full outer electron level • Families have the same number of valence electrons. • As you move across the Periodic Table the number of valence electrons increases. • As you move across the Periodic Table the number of protons increases. • The number of energy levels relates to the period it is located. Period 1 = 1 energy level, Period 2 = 2 energy levels. • The number of valence electrons is the same for each Family