Free Radical Substitution: Chlorination of Methane
Explore the principle, reaction type, and mechanism of free radical substitution in the chlorination of methane. Learn about the overall reaction equation, conditions, and further substitutions.

Free Radical Substitution: Chlorination of Methane
E N D
Presentation Transcript
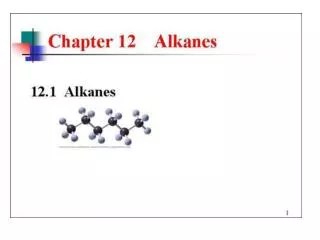
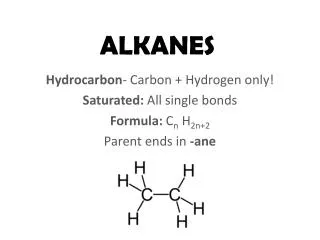
Alkanes Principle reaction type: free radical substitution Mechanism: (chain reaction) substitution
Free radical substitution chlorination of methane i.e. homolytic breaking of covalent bonds Overall reaction equation CH4 + Cl2 CH3Cl + HCl Conditions ultra violet light excess methane to reduce further substitution
Free radical substitution mechanism Cl Cl Cl Cl H Cl H3C H3C H3C H3C Cl Cl Cl H3C H Cl Cl H3C Cl H3CCl H3CCH3 CH3 ultra-violet initiation step two propagation steps termination step minor termination step
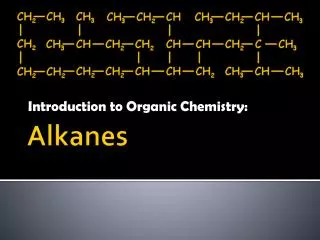
Further free radical substitutions Overall reaction equations CH3Cl + Cl2 CH2Cl2 + HCl CH2Cl2 + Cl2 CHCl3 + HCl CHCl3 + Cl2 CCl4 + HCl Conditions ultra-violet light excess chlorine