C-H Functionalization: Shilov Chemistry
120 likes | 575 Vues
C-H Functionalization: Shilov Chemistry. Allegra Liberman-Martin, Levi Moore, Drexel Proctor, and Giselle Sullivan Chem 165: Organometallic Chemistry Fall 2009. Methane Utilization. Current methane conversion methods are indirect Synthesis gas method

C-H Functionalization: Shilov Chemistry
E N D
Presentation Transcript
C-H Functionalization:Shilov Chemistry Allegra Liberman-Martin, Levi Moore, Drexel Proctor, and Giselle Sullivan Chem 165: Organometallic Chemistry Fall 2009
Methane Utilization • Current methane conversion methods are indirect • Synthesis gas method • Goal: Direct conversion of methane to methanol • Challenging aspect: increasing reactivity of oxidation products Comparing Bond Dissociation Energies (BDEs): BDE(C-H, methane) = 104 kcal/mol BDE(C-H, methanol) = 93 kcal/mol
Radical Chain Oxidation of Alkanes vs. Oxenoid Mechanism Radical Chain Oxidation RH + O2 R• + HOO• R• + O2 ROO• ROO• + RH ROOH + R• • Dioxygen causes initiation and propagation of chains. ROOH + Mn Mn+1 + RO- + •OH • Compounds of transition metals can also participate in the propagation of radicals. The radical mechanism is a 1e- process. Oxenoid Mechanism RH + 2e- + 2H+ + O2 ROH + H2O • Hydroxylation of C-H bond of a hydrocarbon coupled with oxidation of electron donors. • Requires interaction with a metal core, either in a synthesized catalyst or an enzyme. • The oxenoid mechanism is a 2e- process, and does not involve the formation of radicals. • Both major systems currently in use proceed through an oxenoid type mechanism. Muradov, N. Z.; Shilov, A. E.; Shteinman, A. A., Reac. Kin. and Cat.Lett.1975, 2, 417-423. Shilov, A.E.; Shteinman A.A. Acc. Chem. Res.1999, 32, 763, 771.
Shilov System First transition metal system for alkane functionalization Catalytic in Pt(II), but requires stoichiometric Pt(IV) Because methyl undergoes nucleophilic attack by both water and chloride ions in solution, both CH3OH and CH3Cl are generated Luinstra, G. A.; Labinger, J. A.; Bercaw, J. E. J. Am. Chem. Soc. 1993, 115, 3004-3005. Lunistra, G. A.; Wang, L.; Stahl, S. S.; Labinger, J. A.; Bercaw, J. E. J. Organomet. Chem.1995, 504, 75-91.
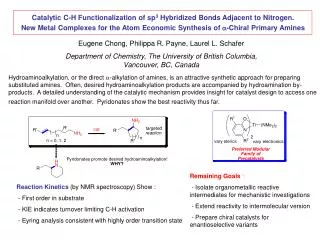
Shilov System - Rate Determining Step • Two steps in question: • Methane uptake • methane platinum complex precursor for C–H • C-H activation • Rate Determining Step: methane uptake • Associative vs. dissociative pathway • Ligand dependency (see respective rate laws) Zhu, H.; Ziegler, T. J. Organomet. Chem. 2006. 691, 4486.
Shilov System: Methane Uptake Associative mechanism for methane uptake Dissociative mechanism for methane uptake Zhu, H.; Ziegler, T. J. Organomet. Chem. 2006. 691, 4486.
Catalytica Systems • 81% selectivity for methyl bisulfate • 89% conversion of methane • 85% selectivity for methyl bisulfate • 50% conversion of methane In both cases, an additional step to hydrolyze methyl bisulfate to methanol is necessary. Periana, R. A.; Taube, D. J.; Evitt, E. R.; Loffler, D. G; Wentreck, P. R.; Voss, G.; Masuda, T. Science1993, 259, 340-343. Periana, R. A.; Taube, D. J.; Gamble, S.; Taube, H.; Satoh, T.; Fuhii, H. Science1998, 280, 560-564.
Vanadate Catalysis • Vanadate/pyrazine-2-carboxylic acid catalysis • Less extreme reaction conditions • Industrial impact for C-H functionalization • Forms methanol, formic and acetic acid Süss-Fink, G.; Stanislas, S.: Shul'pin, G.B.; Nizova, G.V.Appl. Organomet. Chem.2000,14, 623-628.
Gold in the Shilov Reaction System • Au(I) is isoelectronic with Hg(II); Au(III) with Pt(II) • Selenic acid (H2SeO4) used as oxidant, rather than Pt(IV) • Methane converts to methyl bisulfate, which is hydrolyzable to methanol De Vos, D. E.; Sels, B. F. . Angew. Chem. Int. Ed. 2004, 44, 30-32.
Construction of MOF • Wanted to construct a heterogeneous catalyst for Shilov reaction • Used Gd and Pt(BPDC)Cl2 in MOF • Catalytic site structure retained Image (Figure 1, p. 2057) from “Design, synthesis, and characterization of a Pt-Gd metal-organic framework containing potentially catalytically active sites.” Szeto, K. C.; Kongshaug, K. O.; Jakobsen, S.; Tiset, M.; and Lillerud, K. P. Royal Soc. Chem.2008,2054-2060. Szeto, K. C.; Kongshaug, K. O.; Jakobsen, S.; Tiset, M.; and Lillerud, K. P. Royal Soc. Chem.2008, 2054-60.
Formation of Lactones • Best with typical Shilov Pt(II) catalyst and Pt(IV) oxidant (60%yield) • Second best with Cu(II) oxidant (56%yield with 3 eq.) • Can be monomers for organic synthesis, materials science, polymers, or biological molecules Lee, J. M., and Chang, S. Tetra. Lett. 2006, 47, 1375-1379.