Rate Laws
Rate Laws. Text p. 580-585 Topic 16.1.1-16.1.3. Rate Law. An equation that gives its rate as a function of reactant concentrations. Allows chemists to propose reaction mechanisms. Each reaction has its own The general form of the rate law is Rate = k[A] x [B] y k is the rate constant

Rate Laws
E N D
Presentation Transcript
Rate Laws • Text p. 580-585 • Topic 16.1.1-16.1.3
Rate Law • An equation that gives its rate as a function of reactant concentrations. • Allows chemists to propose reaction mechanisms. • Each reaction has its own • The general form of the rate law is Rate = k[A]x[B]y • k is the rate constant • Constant of proportionality • Varies greatly with temperature • For solids, it varies with particle size • Its units will vary • [A]and [B]are the concentrations of the reactants. • x and y are exponents known as rate orders • must be determined experimentally • Typically small whole numbers (0, 1, 2) • x is the order of the reaction with respect to A • y is the order of the reaction with respect to B • Overall order of the reaction is x +y • Expresses how the reaction changes with time. 2.
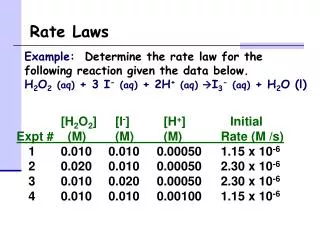
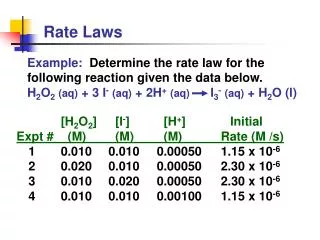
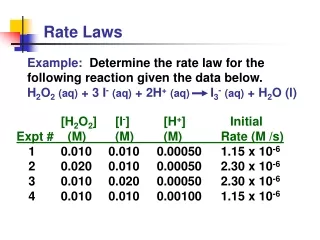
Example 1 NH4+ (aq) + NO2- (aq) N2 (g) + 2H2O (l) Compare Experiments 1 and 2: when [NH4+] doubles, the initial rate doubles. 3.
Example 1 NH4+ (aq) + NO2- (aq) N2 (g) + 2H2O (l) Likewise, compare Experiments 5 and 6: when [NO2-] doubles, the initial rate doubles. 4.
Concentration and Rate NH4+ (aq) + NO2- (aq) N2 (g) + 2H2O (l) This equation is called the rate law, andk is the rate constant. 5.
Example 1 • Solve for k
Example 1 What is the rate of the reaction when[ NH4+ ] is 0.100M and [NO2- ] is 0.100 M?
The Rate Law • Exponents tell the order of the reaction with respect to each reactant. • This reaction is First-order in [NH4+] First-order in [NO2−] • The overall reaction order can be found by adding the exponents on the reactants in the rate law. • This reaction is second-order overall. Rate = K [NH4+]1[NO2- ]1 8.
Determining x, y • Rate = k[A]x[B]y • If x is 1 • doubling [A] will double the rate of the reaction (21) • tripling [A] will triple the rate of the reaction (31) • quadrupling [A] will quadruple the rate of the reaction (41) • If x is 2 • doubling [A] will quadruple the rate of the reaction (22) • tripling [A] will make the reaction 9 times faster (32) • quadrupling [A] will make the reaction 16 times faster (42)
Units of k Depend on Overall Reaction Order • Rate = k[A]x[B]y • k= Rate [A]x[B]y • k= M/s (M/s)x (M/s)y • For a reaction that is second order overall (x + y) = 2 , • k= M/s (M/s)1 (M/s)1 • k= 1 (M/s)1
[NO] mol dm-3 [O2] mol dm-3 Rate mol dm-3 s-1 0.40 0.50 1.6 x 10-3 0.40 0.25 8.0 x 10-4 0.20 0.25 2.0 x 10-4 Determining the Rate constant and Order The following data was collected for the reaction of substances A and B to produce products C and D. a. Deduce the order of this reaction with respect to A and to b. Write an expression for the rate law in this reaction and calculate the value of the rate constant. 11.
The First Order Rate Equation Consider a simple 1st order reaction: A B Rate = k[A] • How much reactant A is left after time t? • How much time is required for a given • amount of A to react? • The rate equation as a function of time can be written as Where [A]t = the concentration of reactant A at time t [A]o = the concentration of reactant A at time t = 0 K = the rate constant Ln [A]t - Ln[A]o = - kt Or Ln[A]t = -kt Ln[A]0 12.
CH3NC CH3CN First-Order Processes Consider the process in which methyl isonitrile is converted to acetonitrile. How do we know this is a first order reaction? 13.
CH3NC CH3CN First-Order Processes This data was collected for this reaction at 198.9°C. Does rate=k[CH3NC] for all time intervals? 14.
First-Order Processes • When Ln P is plotted as a function of time, a straight line results. • The process is first-order. • k is the negative slope: 5.1 10-5 s-1. 15.
Half-Life of a Reaction • Half-life is defined as the time required for one-half of a reactant to react. • Convenient way to describe how fast a reaction will occur. • Because [A] at t1/2 is one-half of the original [A], [A]t = 0.5 [A]0. . 16.
First Order Rate Calculation Example 1: The decomposition of compound A is first order. If the initial [A]0 = 0.80 mol dm-3. and the rate constant is 0.010 s-1, what is the concentration of [A] after 90 seconds? 18.
First Order Rate Calculation Example 1: The decomposition of compound A is first order. If the initial [A]0 = 0.80 mol dm-3. and the rate constant is 0.010 s-1, what is the concentration of [A] after 90 seconds? Ln[A]t – Ln[A]o = -kt Ln[A]t – Ln[0.80] = - (0.010 s-1 )(90 s) Ln[A]t = - (0.010 s-1 )(90 s) + Ln[0.80] Ln[A]t = -0.90 - 0.2231 Ln[A]t = -1.1231 [A]t = 0.325 mol dm-3 19.
First Order Rate Calculations Example 2:A certain first order chemical reaction required 120 seconds for the concentration of the reactant to drop from 2.00 M to 1.00 M. Find the rate constant and the concentration of reactant [A] after 80 seconds. 20.
First Order Rate Calculations Example 2:A certain first order chemical reaction required 120 seconds for the concentration of the reactant to drop from 2.00 M to 1.00 M. Find the rate constant and the concentration of reactant [A] after 80 seconds. Solution k =0.693/t1/2 =0.693/120s =0.005775 s-1 Ln[A] – Ln(2.00) = -0.005775 s-1 (80 s)= -0.462 Ln A = - 0.462 + 0.693 = 0.231 A = 1.26 mol dm-3 21.
First Order Rate Calculations Example 3: Radioactive decay is also a first order process. Strontium 90 is a radioactive isotope with a half-life of 28.8 years. If some strontium 90 were accidentally released, how long would it take for its concentration to fall to 1% of its original concentration? 22.
First Order Rate Calculations Example 3: Radioactive decay is also a first order process. Strontium 90 is a radioactive isotope with a half-life of 28.8 years. If some strontium 90 were accidentally released, how long would it take for its concentration to fall to 1% of its original concentration? Original concentration (100 M, final concentration 1 M) Solution k =0.693/t1/2 =0.693/28.8 yr =0.02406 yr-1 Ln[1] – Ln(100) = - (0.02406 yr-1)t = - 4.065 t = - 4.062 . - 0.02406 yr-1 t = 168.8 years 23.
Second-Order Processes Similarly, integrating the rate law for a process that is second-order in reactant A: Rate = k[A]2 1 [A]o 1 [A]t = kt + So if a process is second-order in A, a graph of 1/[A] vs. twill yield a straight line with a slope of k. 24.
NO2(g) NO (g) + 1/2 O2(g) Determining Reaction OrderDistinguishing Between 1st and 2nd Order The decomposition of NO2 at 300°C is described by the equation: A experiment with this reaction yields this data: 25.
Determining Reaction OrderDistinguishing Between 1st and 2nd Order Graphing ln [NO2] vs.t yields: • The graph is not a straight line, so this process cannot be first-order in [A]. Does not fit the first order equation: 26.
Second-Order Reaction Kinetics A graph of 1/[NO2] vs. t gives this plot. • This is a straight line. Therefore, the process is second-order in [NO2]. • The slope of the line is the rate constant, k. 27.
Half-Life for 2nd Order Reactions In this case the half-life depends on the initial concentration of the reactant A. 28.
Sample Problem 1: Second Order Acetaldehyde, CH3CHO, decomposes by second-order kinetics with a rate constant of 0.334 mol-1dm3s-1 at 500oC. Calculate the amount of time it would take for 80% of the acetaldehyde to decompose in a sample that has an initial concentration of 0.00750 M. 29.
Sample Problem 1: Second Order Acetaldehyde, CH3CHO, decomposes by second-order kinetics with a rate constant of 0.334 mol-1dm3s-1 at 500oC. Calculate the amount of time it would take for 80% of the acetaldehyde to decompose in a sample that has an initial concentration of 0.00750 M. The final concentration will be 20% of the original 0.00750 M or = 0.00150 1 . .00150 1 . .00750 = 0.334 mol-1dm3s-1t + 666.7 = 0.334 t + 133.33 0.334 t = 533.4 t = 1600 seconds 30.
Sample Problem 2: Second Order Acetaldehyde, CH3CHO, decomposes by second-order kinetics with a rate constant of 0.334 mol-1dm3s-1 at 500oC. If the initial concentration of acetaldehyde is 0.00200 M. Find the concentration after 20 minutes (1200 seconds) Solution 31.
Sample Problem 2: Second Order Acetaldehyde, CH3CHO, decomposes by second-order kinetics with a rate constant of 0.334 mol-1dm3s-1 at 500oC. If the initial concentration of acetaldehyde is 0.00200 M. Find the concentration after 20 minutes (1200 seconds) Solution 1 . [A]t 1 . 0.00200 mol dm-3 = 0.334 mol-1dm3s-1(1200s) + 1 . [A]t = 0.334 mol-1dm3 s-1(1200s) + 500 mol-1dm3 = 900.8 mol-1dm3 1 _____. 900.8 mol-1dm3 [A]t = = 0.00111 moldm-3 32.
Homework • Read p. 580-591 • Problems p. 619-620 • # 14.37-14.46
Collision Model • Topic 6.2.1 - 6.2.7 • Topic 16.3.1-16.3.2 • Text p. 591-597, p. 605-612
The Collision Model • Chemical reaction • bonds broken bonds formed • Molecules can only react if they collide with • sufficient energy • appropriate orientation. 36.
Activation Energy (Ea) • Minimum amount of energy required for reaction 37.
Reaction Coordinate Diagrams It is helpful to visualize energy changes throughout a process on a reaction coordinate diagram like this one for the rearrangement of methyl isonitrile. 38.
Reaction Coordinate Diagrams • Shows energy of reactants and products (and E). • High point diagram is transition state. • Species present the transition state is called activated complex. • Energy gap between reactants and activated complex is activation energy barrier. 39.
Temperature and Rate • Reaction rate increases as temperature increases. • k is temperature dependent. • Reaction rate increases about 10 fold for each 10oC rise in temperature 40.
Maxwell–Boltzmann Distributions • Temperature-- a measure of the average kinetic energy of the molecules in a sample. • At any temperature there is a wide distribution of kinetic energies. 41.
Maxwell–Boltzmann Distributions • As temperature increases, the curve flattens and broadens. • At higher temperatures, more molecules have higher energy. 42.
Maxwell–Boltzmann Distributions • If the dotted line represents the activation energy, at higher temps the fraction of molecules that can overcome the activation energy barrier increases. • As a result, the reaction rate increases. 43.
Maxwell–Boltzmann Distributions This fraction of molecules can be found through the expression: where R is the gas constant and T is the temperature in Kelvin . 44.
Example • Ea = 100 KJ/mol • T = 300 K • R = 8.314 J/Mol K • f= 3.9 X10-18 • Ea = 100 kJ/mol • T = 310 K • R = 8.314 J/Mol K • f = 1.4 X 10-17 • There are 3.6 times more molecules possessing the activation energy.
Svante Arrhenius • Swedish Chemist (1888) • Noticed that increase in rate constant with increase in temp is NOT linear. • Determined that reaction data obeyed an equation based on • Fraction of molecules with Ea or greater • # of collisions per second • Fraction of collisions with appropriate orientation
Arrhenius Equation Arrhenius developed a mathematical relationship between k and Ea: A --frequency factor represents the likelihood that collisions would occur with the proper orientation for reaction. Ea --activation energy. T --Kelvin temperature R --universal thermodynamics (gas) constant. R = 8.314 J mol-1 K-1 47.
1 RT Arrhenius Equation Taking the natural logarithm of both sides, the equation becomes y = mx + b When k is determined experimentally at several temperatures,Eacan be calculated from the slope of a plot of ln k vs. 1/T. 48.
Assignment • Read p. 591-597, 605-612 • P. 620 14.49-14.54,14.57, 14.58 • Read Handout—”Factors affecting the rate of a reaction”
Reaction Mechanisms • Topic 16.2.1-16.2.2 • Textbook p. 597-604