Reversible and irreversible Processes
250 likes | 1.37k Vues
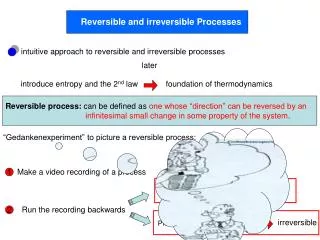
Reversible and irreversible Processes. intuitive approach to reversible and irreversible processes . later. introduce entropy and the 2 nd law . foundation of thermodynamics. Reversible process: can be defined as one whose “direction” can be reversed by an

Reversible and irreversible Processes
E N D
Presentation Transcript
Reversible and irreversible Processes intuitive approach to reversible and irreversible processes later introduce entropy and the 2nd law foundation of thermodynamics Reversible process: can be defined as one whose “direction” can be reversed by an infinitesimal small change in some property of the system. “Gedankenexperiment” to picture a reversible process: Make a video recording of a process 1 reversible Observable process Run the recording backwards 2 irreversible Process impossible to observe
reversible Examples: Process is possible reversible Backward recording Backward recording
x x reversible irreversible Small changes can be reversed but V1 ,Ts V2 ,Tf gas You never observe reversed process of free expansion
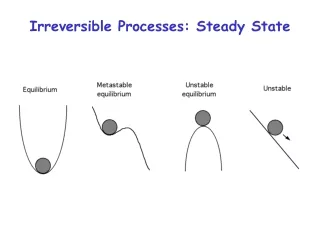
(in strictest sense, almost all real processes are irreversible) Reversibility is an idealization Reversibility requires equilibrium processes but Not every equilibrium process is reversible Almost perfect insulation Example Tg gas in equilibrium at any time > T0 Qout Although system in equilibrium, no small change of the system will reverse the heat flow
Reversibility is an idealization Dry friction between 2 objects You never observe the reversed process: object starts to move without assistance
x Heat Conductivity in Solids (an example for irreversibility) Friction between piston and cylinder irreversibility Remember: Heat is an energy transferred from one system to another because of temperature difference T1 > T2 System 2 System 1 Heat Q flows from 1to2
T1 > T2 Heat reservoir 2 Heat reservoir 1 *(in the textbook T2>T1) L T(x) T1 T2 x 0 L A Heat transfer per time interval through homogeneous solid object: K: thermal conductivity of the rod L where A: cross-section of the rod
Electric Systems (examples for reversibility and irreversibility) #1 + battery - : work done against electrical forces per unit charge Work: Current
#1 #2 #1 1. law: #2 No heat transferred (Q=0) Irreversible case Resistor network with total resistance R In the steady state: Internal energy of black box unchanged Heat leaving the system Application as heater reversible case Capacitor network with total Capacity C - Charging an uncharged capacitor - Discharge of the capacitor done by the capacitor









![A] reversible B] irreversible](https://cdn2.slideserve.com/4685684/slide1-dt.jpg)