MOISTURE AND TOTAL SOLID ANALYSIS
MOISTURE AND TOTAL SOLID ANALYSIS. SFA3023 Food Analysis. Importance of Moisture Assay. Legal and Labeling Requirements Economic Microbial stability Food Quality Food Processing Operations. Forms of Water in Foods. Free water Water retains its physical properties Capillary water

MOISTURE AND TOTAL SOLID ANALYSIS
E N D
Presentation Transcript
MOISTURE AND TOTAL SOLID ANALYSIS SFA3023 Food Analysis
Importance of Moisture Assay • Legal and Labeling Requirements • Economic • Microbial stability • Food Quality • Food Processing Operations
Forms of Water in Foods • Free water • Water retains its physical properties • Capillary water • Water is held tightly within spaces within a food that are surrounded by a physical barrier • E.g. An emulsion droplet • Bound water • Water is bound physically (e.g. in protein) or chemically (e.g. Na2SO4. 10H2O)
Determination of Moisture: Methods • Drying Methods • Oven drying methods • Distillation method • Dean and Stark Method • Chemical Methods • Karl Fisher • Gas production • Physical Methods
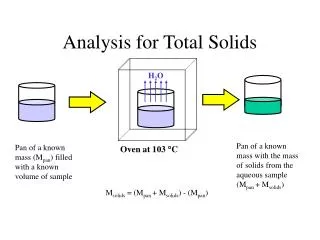
Drying Methods • These methods rely on measuring the mass of water in a known mass of sample. • The moisture content is determined by measuring the mass of a food before and after the water is removed by drying. • Basic Principle • Water has a lower boiling point than the other major components within foods such as lipids, protein and carbohydrate.
Sometimes, moisture content also reported as “total solid” • Total solid is a measure of the amount of material remaining after all the water has been evaporated • Advantages • Cheap, easy to use, many samples can be analyzed simultaneously • Disadvantages • Destructive, time consuming
Moisture and total solids contents of food can be calculated using one of the equations below: • % Moisture (wt/wt) = wt H2O in sample X 100 wt of wet sample • % Moisture (wt/wt) = wt of wet sample - wt of dry sample X 100 wt of wet sample • % Total solids (wt/wt) = wt of dry sample X 100 wt of wet sample
Types of Oven • Convection Oven • Greatest temperature variations - because hot air slowly circulated with out the aid of fan, air movement is obstructed further by pans placed in the oven
2. Forced Draft Oven • The least temperature differential across the interior (< 1oC). Air is circulated by a fan that forces air movement throughout the oven cavity • Drying period 0.75 – 24 hr, depending on food sample and its pretreatment
Vacuum oven • Drying under reduced pressure (25 – 100mm Hg). • Able to obtain a more complete removal of water and volatiles without decomposition within a 3 – 6 hr
4. Microwave Oven • Weighed samples are placed in a microwave oven for a specified time and power-level and their dried mass is weighed.
In microwave oven, water evaporation is due to absorption of microwave energy, which causes them to become thermally excited. Advantage simple Disadvantage: Care must be taken to standardize the drying procedure and ensure that the microwave energy is applied evenly across the sample.
5. Infrared lamp drying • Principle of drying: Similar to microwave oven • Advantages: rapid and inexpensive • This is because the IR energy penetrates into the sample
To produce consistent results, one must control • The distance between the sample and IR lamp • The dimensions of the samples • IR drying methods are not officially recognized for moisture content determinations because it is difficult to standardize the procedure.
6. Moisture Analyzer • Using a digital balance, the test sample is placed on an aluminum pan and the constant temperature is applied to the test sample. • Instrument automatically weighs and calculates the % of moisture or solids
Practical Considerations • Sample Dimensions • Surface area of material exposed to the environment is important • Clumping and surface crust formation • Mix the sample with dried sand to prevent clumping • Decomposition of other food components • High temp or too long drying time can cause decomposition of some heat-sensitive components in the food
Volatilization of other food components • Food with high volatile components should be analyzed using distillation or chemical method. • High moisture samples • Dried in two stages to prevent spattering • Sample pans • Should be dried in the oven and stored in a dessicator prior analysis • Avoid contact with bare hands. Use tongs or cotton gloves
Type of water • free vs. bound water • Example: % water in milk vs. non-fat dried milk. • Temperature and power level variations • Significant variations in temperature and power levels within an drying instruments.
Distillation Methods • Direct measurement of the amount of water removed from a food sample by evaporation • Involve co-distilling the moisture in a food sample with a high boiling point solvent that is immiscible in water, collecting the mixture that distills off and then measuring the volume of water
Advantages: • Suitable for low moisture foods and foods containing volatile oils such as herbs and spices • Cheap, easy to set up and operate • Disadvantage: • Destructive, time consuming, involve flammable solvent, not applicable for some types of foods
Dean and Stark Method • A known weight of sample is placed in a flask with immiscible organic solvent such as xylene or toluene. • The flask containing the sample and organic solvent is attached to a condenser by a side arm and the mixture is heated • The water in the sample evaporates and moves up to the condenser • Liquid water will be collected in graduated tube • When no more water collected, distillation is stopped.
Practical Consideration • There are a number of practical factors that can lead to erroneous results: • Emulsions can sometimes form between the water and the solvent which are difficult to separate • Water droplets can adhere to the inside of the glassware • Decomposition of thermally labile samples can occur at the elevated temperatures used.
Chemical Methods • Moisture is determined by the reactions between water and certain chemical reagents • A chemical reagent is added to the food that reacts specifically with water to produce a measurable change in the properties of the system, e.g., mass, volume, pressure, pH, color, conductivity. • Type of chemical method commonly used: • Karl Fischer Titration • Gas production Methods
Measurable changes in the system are correlated to the moisture content using calibration curves. • For accurate measurement, chemical reagent must reacts with all of the water molecules present, but not with any of the other components in the food matrix.
Chemical reaction do not involve application of heat. • Therefore, they are suitable for: • Foods that contain thermally labile substances that would change the mass of the food matrix on heating (e.g., food containing high sugar concentrations) or • Foods that contain volatile components that might be lost by heating (e.g. spices and herbs).
Karl-Fischer Titration • Determine the low moisture foods (e.g. dried fruits and vegetables, confectionary, coffee, oils and fats) or low moisture food high in sugar or protein. • It is based on the following reaction: 2H2O + SO2 + I2→ H2SO4 + 2HI
Procedure • The food to be analyzed is placed in a beaker containing solvent and is then titrated with Karl Fisher reagent (a solution that contains iodine). • Iodine will reacts with remaining water in the samples. • The endpoint color is dark red-brown. • The volume of iodine solution required to titrate the water is measured and can be related to the moisture content using a pre-prepared calibration curve.
Major Difficulties and Source of Error • Incomplete water extraction • Fineness of grind is important • Atmospheric moisture • External air must not be allowed to infiltrate the reaction chamber • Moisture adhering to walls of unit • All glassware and utensils must be carefully dried • Interferences from certain food constituents • Oxidation of ascorbic acid to dehydroascorbic acid
Gas Production Methods • Commercial instruments are also available that utilize specific reactions between chemical reagents and water that lead to the production of a gas • Example; when a food sample is mixed with powdered calcium carbide, the amount of acetylene gas produced is related to the moisture content.
The amount of gas produced can be measured by • The volume of the gas produced • The decrease in the mass of the sample after the gas is released • The increase in pressure of a closed vessel containing the reactants
Physical Methods • Electrical methods • Dielectric Method • Moisture is determined by measuring the change in capacitance or resistance to an electric current passed through the sample • Limited to food contains not more than 30-35% moisture.
Conductivity method • The conductivity of an electric current increases with the percentage of moisture sample • Must keep the temperature constant
2. Hydrometry • Measuring specific gravity or density • Best applied to the analysis of solutions consisting of only one component in a medium of water • Commonly used in beverages, salt brines and sugar solutions • Example: Pycnometer, hydrometer, Westphal Balance.
3. Refractometry • Determine the soluble solids in fruits and fruit products • Rapid and accurate methods • Principle: when a beam of light is passed from one medium to another and the density differs, then the beam is bent or refracted.
4. Infrared Analysis • Principle: • Measure absorption of radiation by molecules in foods • Different functional groups absorb different frequencies of infrared radiation • For water, near-infrared (NIR) bands (1400-1450, 1920-1950 nm) are characteristic of the –OH stretch of water molecule
Methods to determine Water in Different Molecular Environments • Vapour Pressure Methods • Determination of water activity • Bound water is less volatile than free water • Thermogravimetric Methods • Measure the mass of a sample as it is heated at a controlled rate • Free water normally evaporates at a lower temperature than bound water
Calorimetric Methods • Using Differential Scanning Calorimetry (DSC) and Differential Thermal Analysis (DTA) • Measure changes in the heat absorbed or released by a material as its temperature is varied at a controlled rate.
Spectroscopic Methods • Nuclear magnetic resonance (NMR) – distinguish molecules within materials based on their molecular mobility • Molecular mobility for free water is higher than that of bound water
Comparison of the Methods • Oven drying methods: involve the removal of moisture from the sample and then a weight determination of the solids remaining. • Distillation methods: Involve a separation of the moisture from the solids. The moisture content is calculated directly by volume. • Chemical Methods: reflected as the amount of titrant used.
Dielectric and conductivity methods: electrical properties of water • Hydrometric methods: based on the relationship between specific gravity and moisture content • Refractive Index: how water in a sample affects the refraction of light • NIR Methods: absorption at w/length characteristic of the molecular vibration in water.
Nature of Sample • Oven Drying: problem with volatile compounds and chemical degradation • Distillation: minimize volatilization and decomposition • Karl Fischer: Food with very low moisture or high in fats and sugars • Pycnometer, hydrometer and refractometer requires liquid samples with limited constituents
Intended Purposes • Rapid analysis but less accurate: oven drying methods, microwave drying, infrared drying and moisture analyzer • Rapid but require correlation to less empirical methods: electrical, hydrometric, refractive index and infrared analysis methods
Summary • Types of water present: free, adsorbed and hydration of water. • Major difficulty in many methods is attempting to remove or otherwise quantitate all water present. • Factors to be considered in selecting moisture analysis: • expected moisture content, • nature of the other food constituents (i.e. highly volatile), • equipment availability, • speed necessary, • accuracy and precision required and • intended purpose (e.g. regulatory or in-plant control)