Cucurbituril
Cucurbituril.

Cucurbituril
E N D
Presentation Transcript
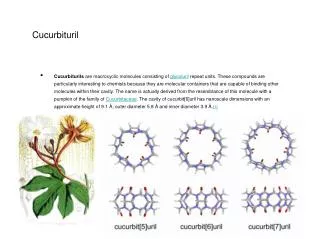
Cucurbituril • Cucurbiturils are macrocyclic molecules consisting of glycoluril repeat units. These compounds are particularly interesting to chemists because they are molecular containers that are capable of binding other molecules within their cavity. The name is actually derived from the resemblance of this molecule with a pumpkin of the family of Cucurbitaceae. The cavity of cucurbit[6]uril has nanoscale dimensions with an approximate height of 9.1 Å, outer diameter 5.8 Å and inner diameter 3.9 Å.[1]
Cucurbiturils were first synthesized in 1905 by Behrend[2] but their structure was not elucidated until 1981.[3] To date cucurbiturils composed of 5, 6, 7, 8, and 10 repeat units have all been isolated,3 which have internal cavity volumes of 82, 164, 279, 479, and 870 Å2 respectively. A cucurbituril composed of 9 repeat units has yet to be isolated. Other common molecular capsules that share a similar molecular shape with cucurbiturils include cyclodextrins and calixarenes.
Host-Guest chemistry • Cucurbiturils are efficient host molecules in molecular recognition and have a particularly high affinity for positively charged or cationic compounds. High association constants with positively charged molecules are attributed to the carbonyl groups that line each end of the cavity and can interact with cations is a similar fashion to crown ethers. The affinity of cucurbiturils can be very high. For example the affinity equilibrium constant of cururbit[7]uril with the positively charged 1-aminoadamantane hydrochloride is experimentally determined at 4.23*1012.[6] • Host guest interactions also significantly influence solubility behavior of cucurbiturils. Cucurbit[6]uril dissolves poorly in just about any solvent but solubility is greatly improved in a solution of potassium hydroxide or in an acidic solution. The cavitand forms a positively charged inclusion compound with a potassium ion or a hydronium ion respectively which have much greater solubility that the uncomplexed neutral molecule.[7]
Applications • Given their high affinities to form inclusion complexes cucurbiturils have been employed as the macrocycles component of a rotaxane. After formation of the supramolecular assembly or threaded complex with a guest molecule such as hexamethylene diamine the two ends of the guest can be reacted with bulky groups that will then act as a stoppers preventing the two separate molecules from dissociating.[8] • In another rotaxane system with a CB[7] wheel, the axle is a 4,4'-bipyridinium or viologen subunit with two carboxylic acid terminated aliphatic N-substituents at both ends.[9] In water at concentration higher than 0.5 M complexation is quantitative without need of stoppers. At pH = 2 the carboxylic end-groups are protonated and the wheel shuttles back and forth between them as evidenced by the presence of just two aromatic viologen protons in the proton NMR spectrum. At pH = 9 the wheel is locked around the viologen center. • Cucurbiturils host-guest properties have been explored for drug delivery vehicles. The potential of this application has been explored with cucurbit[7]uril that forms an inclusion compound with the important cancer fighting drug oxaliplatin. CB[7] was employed despite the fact that it is more difficult to isolate since it has much greater solubility in water and its larger cavity size can accommodate the drug molecule. The resulting complex was found to have increased stability and greater selectivity that may lead to less side effects.[10]
Applications • Cucurbiturils have also been explored as supramolecular catalysts. Larger cucurbiturils, such as cucurbit[8]uril can bind multiple guest molecules. CB[8] forms a complex 2:1 (guest:host) with (E)-diaminostilbene dihydrochloride which is accommodated by CB[8]’s larger internal diameter of 8.8angstrom and height 9.1angstrom.[11] The close proximity and optimal orientation of the guest molecules within the cavity enhances the rate of thephotochemical cyclization to givecyclobutanedimer with a 19:1 stereoselectivity for the syn configuration when bound to CB[8]. In the absence of CB[8] the cyclization reaction does not occur, but only the isomerization of thetrans isomer to thecis isomer is observed.[12] • CB[10] can is large enough to hold other molecular hosts such as a calixarene molecule. With a calixarene guest different chemical conformations (cone, 1,2-alternate, 1,3-alternate) are in rapid equilibrium. Allosteric control is provided when an adamantane molecule forces a cone conformation with a calixarene - adamantane inclusion complex within a CB[10] molecule.
References • Review: The Cucurbit[n]uril Family Jason Lagona, Pritam Mukhopadhyay, Sriparna Chakrabarti, Lyle IsaacsAngewandte Chemie International Edition Volume 44, Issue 31, Pages 4844 - 4870 2005 • Behrend et al Liebigs Ann. Chem. 1905, 339, 1. • Cucurbituril W. A. Freeman, W. L. Mock, and N.-Y. Shih J. Am. Chem. Soc., 1981, 103, 7367. • Cucurbituril Homologues and Derivatives: New Opportunities in Supramolecular Chemistry Acc. Chem. Res., 36 (8), 621 -630, 2003. • Cucurbit[10]uril Simin Liu, Peter Y. Zavalij, and Lyle Isaacs J. Am. Chem. Soc.; 2005; 127(48) pp 16798 - 16799; (Communication) DOI: 10.1021/ja056287n • The Cucurbit[n]uril Family: Prime Components for Self-Sorting Systems Simin Liu, Christian Ruspic, Pritam Mukhopadhyay, Sriparna Chakrabarti, Peter Y. Zavalij, and Lyle Isaacs J. Am. Chem. Soc.; 2005; 127(45) pp 15959 - 15967. • The complex formation of a, w-dicarboxylic acids and a, w-diols with cucurbituril and a-cyclodextrin. the first step to the formation of rotaxanes and polyrotaxenes of thepolyester type Hans-Jürgen Buschmann, Klaus Jansen, Eckhard Schollmeyer Acta Chim. Slov. 1999, 46(3), pp. 405-411 • Switching a molecular shuttle on and off: simple, pH-controlled pseudorotaxanes based on cucurbit[7]uril Vladimir Sindelar, Serena Silvi, Angel E. Kaifer Chemical Communications, 2006, 2185 - 2187 • Novel molecular drug carrier: encapsulation of oxaliplatin in cucurbit[7]uril and its effects on stability and reactivity of the drug Young Jin Jeon, Soo-Young Kim, Young Ho Ko, Shigeru Sakamoto, Kentaro Yamaguchi and Kimoon Kim Organic & Biomolecular Chemistry, 2005, 3(11), 2122 - 2125 • A facile, stereoselective [2 + 2] photoreaction mediated by cucurbit[8]uril Sang Yong Jon, Young Ho Ko, Sang Hyun Park, Hee-Joon Kim and Kimoon Kim Chemical Communications, 2001, (19), 1938 - 1939 • Template directed photodimerization of trans-1,2-bis(n-pyridyl)ethylenes and stilbazoles in water Mahesh Pattabiraman, Arunkumar Natarajan, Raja Kaliappan, Joel T. Mague and V. Ramamurthy Chemical Communications, 2005, (36), 4542 - 4544 • The Inverted Cucurbit[n]uril Family Isaacs, L.; Park, S.-K.; Liu, S.; Ko, Y. H.; Selvapalam, N.; Kim, Y.; Kim, H.; Zavalij, P. Y.; Kim, G.-H.; Lee, H.-S.; Kim, K. J. Am. Chem. Soc.; (Communication); 2005; 127(51); 18000-18001.