Understanding Carboxyl Group in Organic Acids: Structure and Naming
The carboxyl group (-COOH) is a key functional group in organic chemistry, characterized by a carbon atom double-bonded to an oxygen atom and single-bonded to a hydroxyl group (OH). This functional group is typically found at the end of carbon chains, making it a defining feature of organic acids. In solutions, the hydrogen from the hydroxyl contributes to the acidity. To name organic acids, identify the longest alkane chain, remove the “e”, and add “oic acid”. Common examples include methanoic (formic) acid and ethanoic (acetic) acid.

Understanding Carboxyl Group in Organic Acids: Structure and Naming
E N D
Presentation Transcript
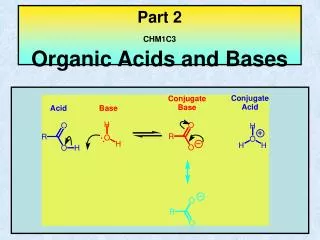
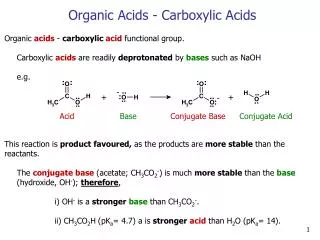
B. Organic Acids Second functional group 1. Carboxyl Group -the functional group is the carboxyl group O || --C--OH Notice how the C is double bonded to one oxygen Since there is only one more bonding site, the carboxyl group is always found on the end of a chain H H H O | | | || H-C--C--C--C--OH | | | H H H - H H+ H+ H+ In solution, it is the H from the OH that is creates the H+ Always written as “COOH” at the end of the molecular formula H H H O | | | || H-C--C--C--C--OH | | | H H H C3 H7 COOH This is how you recognize an organic acid
Practice - Write out the molecular formula for the following O H || | HO-C--C--H | H H H O | | || H-C--C--C--OH | | H H O || H--C--OH CH3COOH HCOOH C2H5COOH 2. Naming Organic Acids Take the longest alkane chain, remove the “e” and add “oic acid” O || H--C--OH Methanoic acid Commonly called Formic acid H O | || H-C--C--OH | H Commonly called Acetic acid Ethanoic acid
Example - Name the following acids H H O | | || H-C--C--C--OH | | H H H H O | | || H-C--C--C--OH | | HCHH | H O H H Cl H || | | | | HO-C--C--C--C--C--H | | | | H H Cl H Propanoic acid Butanoic acid 4,4-dichloro Pentanoic acid