Cutaneous Melanoma
Cutaneous Melanoma. Introduction. melanocytes reside at the dermal/epidermal junction When melanoma arises in the skin, it usually arises from melanocytes at the dermal / epidermal junction . T here are alternate presentations, mucosal melanomas, ocular melanomas,

Cutaneous Melanoma
E N D
Presentation Transcript
Introduction • melanocytes reside at the dermal/epidermal junction • When melanoma arises in the skin, it usually arises from melanocytes at the dermal /epidermal junction .
There are alternate presentations, • mucosal melanomas, • ocular melanomas, • metastatic melanomas from unknown primary sites • presumed primary visceral melanomas. • Each of these has significant differences in presentation and management.
The management of malignant melanoma involves prevention, early diagnosis, surgical extirpation, and combination management of metastatic disease.
Cutaneous Melanoma Biology • The transition from melanocyte to metastatic melanoma involves several histologic intermediates, including : • melanocyticatypia, • atypical melanocytic hyperplasia, • radial growth phase melanoma, • vertical growth phase melanoma, • metastatic melanoma.
The RGP : melanoma in situ (MIS) or superficial invasion into the papillary dermis, or both. • rarely symptomatic • ideal time to diagnose melanoma, • these lesions typically progress to the VGP,
RGP • very low metastatic capacity. • an excellent prognosis • mortality risk at the low end of the 0% to 5% range.
VGP • Risk of melanoma progression is most associated with the presence of VGP, the depth of invasion, and other markers of the malignant phenotype in the VGP component of a melanoma.
the extent of RGP (e.g., clinically, the diameter of the skin lesion) and multiplicity of RGP lesions are not associated with significant risk of metastasis or melanoma-associated mortality
some melanomas are nonpigmented • Others develop a VGP in the absence of a RGP (nodular melanoma histology), • some melanomas present as metastatic melanoma in lymph nodes, skin, subcutaneous tissue, or visceral sites without an apparent primary cutaneous site.
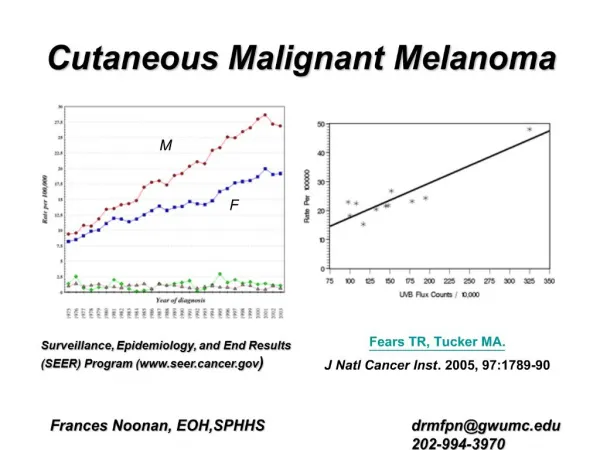
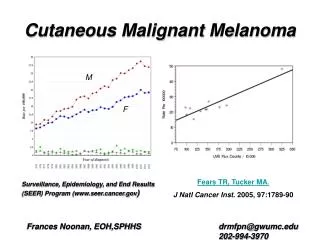
Epidemiology • sixth-most-common U.S. cancer diagnosis • Overall 5-year survival rates for melanoma have increased from 82% in the late 1970s (1975 to 1977) to 92% in the more recent era (1996 to 2002). • a combined effect of ultraviolet (UV) sunlight exposure and fair skin
Ocular and nonacralcutaneous melanomas are 50- to 200-fold more likely in white populations than in nonwhite populations, but melanomas in acral and mucosal sites are within twofold of each other across racial groups.
Recent data suggest significant molecular differences between acral melanomas and melanomas arising on the skin associated with chronic sun damage, with B-RAF and N-RAS mutations in 81% of melanomas on chronically sun-damaged skin, whereas those mutations were uncommon in melanomas from acral or mucosal sites or from skin without chronic sun damage.
Changes in Incidence • An increase in age-adjusted melanoma • A greater proportion of patients diagnosed at earlier and noninvasive stages of disease.
Gender and Age Distribution • the gender ratio of melanoma at diagnosis is approximately 1:1 but is shifting toward a greater proportion of men
Melanoma in Children, Infants, and Neonates • Current recommendations for management of melanoma in children and infants are the same as for adults, and outcomes are generally believed to be comparable.
Anatomic Distribution • The most common sites in males are on the back and in the head and neck regions. In women, the most common sites are in the lower extremities, commonly below the knee. • Lentigomaligna melanoma (LMM) most commonly arises on sun-damaged surfaces of the head and neck in older patients.
Etiology and Risk Factors • ultraviolet light exposure • the relative role of each type of UV irradiation in melanoma etiology is debated. • a combination of fair skin that burns easily and high ultraviolet/sun exposure • The role of sunlight intensity and frequency is debated, but both chronic and intermittent exposure may be relevant
Therapeutic Radiation as a Risk Factor for Melanoma • Radiation doses greater than 15 Gy delivered to pediatric oncology patients has been shown to increase the risk of developing malignant melanoma • In children undergoing scalp irradiation for tineacapitis, scalp basal cell carcinomas were reported to be 30 times more likely to develop than melanomas in adulthood.
Pregnancy and Estrogen Use • no evidence of any negative (or positive) impact of prior, concurrent, or subsequent pregnancy on clinical outcome. • no clear prognostic relevance for birth control pills or estrogen replacement therapy.
Pregnancy • The general recommendation for treatment of women with melanoma diagnosed during pregnancy is to manage them in the same fashion as nonpregnant patients.
Pregnancy • There is no evidence that a subsequent pregnancy adversely impacts outcome. • Measures of the risk of future disease progression can be defined based on the initial prognosis and the subsequent elapsed time without recurrence, and such information may help to guide patients with this challenging question.
Prevention and Screening • Sun Protection • Screening for Early Diagnosis • Self-Examination • Management of the Patient with Numerous Atypical Moles
Sun Protection • Ultraviolet exposure and sunburns, in particular, appear to be etiologic in most melanomas. • there is no formal proof that sunscreens prevent melanoma.
Screening for Early Diagnosis Self-Examination • family members • educating patients • dermatology visits
Management of the Patient with Numerous Atypical Moles • atypical mole syndrom • dysplastic nevus syndrome • B-K mole syndrome. These patients have a heightened risk of melanoma, and this is commonly a familial feature.
Management of the Patient with Numerous Atypical Moles • These patients deserve particular attention to melanoma prevention through sun protection and to early diagnosis through aggressive screening. • routine skin exams by a dermatologist are usually recommended, as often as every 3 months. • dermoscopy(epiluminescent microscopy)
It is tempting to consider excision of all dysplastic nevi. • Melanomas may arise de novo in 30% to 70% of cases, and so it is not clear that removal of all suspicious nevi will lead to a meaningful improvement in survival. • However, it is certainly appropriate to biopsy any nevus that is suspicious, especially one that is changing.
Diagnosis of Primary Melanoma Characteristics of Primary Melanoma • ABCD for asymmetry, border irregularity, color variation, and diameter greater than 6 mm • a wide range in the appearanc • change in a lesion over time or new development of a lesion • ugly duckling sign:amelanotic (nonpigmented) • Being different: amelanotic (nonpigmented)
hallmarks of a late diagnosis • symptomatic, Bleeding Itching pain ulceration • these usually connote deep vertical growth
Biopsy • a full-thickness biopsy of the entire lesion, with a narrow (1 to 2 mm) margin of grossly normal skin. • The depth of excision should include the full thickness of dermis and thus should be within the subcutaneous tissue, • desmoplastic melanoma often arises from lentigomaligna melanoma and is difficult to diagnose both clinically and histologically. Shave biopsies of these lesions can often lead to failure to appreciate the desmoplastic melanoma in the dermis and may substantially delay diagnosis
For some large lesions (e.g., greater than 2 cm diameter) in cosmetically sensitive locations (e.g., face or genitalia), there may be a rationale for an incisional biopsy, but that also should be performed as a full-thickness skin biopsy. Ideally, it should include the most suspicious area of the lesion and also should include if possible a portion of the edge of the lesion, where it transitions to normal skin, to enable assessment of the junctional change. The incisional biopsy may be an elliptical incision or it may be a full-thickness 4- to 6-mm punch biopsy..
Punch biopsies are problematic if too small, if they do not include full-thickness skin, if they are crushed during removal, if they are oriented inaccurately in the paraffin block, or if they are too small to include both the edge of the lesion and the most suspicious or most raised part of the lesion
Melanoma Subtypes: Histologic Growth Patterns • Superficial Spreading Melanoma • Nodular Melanoma • AcralLentiginous Melanoma • LentigoMaligna Melanoma • Lentiginous Melanoma • Desmoplastic Melanoma
Superficial Spreading Melanoma • The most common type • It is typical for the trunk and extremities, except on acral sites. • associated with pagetoid growth of atypical melanocytes in the epidermis. • associated with sun exposure
Nodular Melanoma • Nodular melanomas (NMs) lack a radial growth phase, • may be nonpigmented, • worst prognosis
AcralLentiginous Melanoma • acral sites (subungual, palmar, plantar) and on mucosal surfaces (anorectal, nasopharyngeal, female genital tract). • independent of ultraviolet light exposure • a prolonged radial growth phase before vertical growth • harder to diagnose • DDx subungual hematomas, which can lead to diagnostic delay
LentigoMaligna Melanoma • older individuals • sun-damaged skin,( face). • an extensive radial growth phase • When just melanoma in situ, this radial growth phase portion is called lentigomaligna (LM) or Hutchinson's freckle,
Lentiginous Melanoma • Early radial growth phase melanomas • Over time, this may represent a growing proportion of melanomas that have traditionally been grouped as SSM, LM, ALM, or unclassified melanomas.
Desmoplastic Melanoma • histologically manifested by dermal melanocytes in a dense stromal response • usually nonpigmented • usually stain negative for MART-1/MelanA, gp100, and tyrosinase, but they do stain for S-100. • most commonly in the head and neck • de novo as a nonpigmented skin papule or as a dermal/vertical growth phase component arising from a pre-existing lentigomaligna or other pigmented junctional lesion
Desmoplastic Melanoma • a high rate of local recurrence • the risk of lymph node metastasis is lower • a higher threshold for performing sentinel node biopsy (SNBx) in patients with pure desmoplastic melanoma.
Prognostic Factors for Primary Melanomas • Depth of Invasion • Ulceration • age, • angiolymphatic invasion • mitotic rate • Gender • body site
Depth of Invasion • Clark defined depth based on the layer of skin to which the melanoma has invaded. • Clark level I melanomas are melanomas in situ, limited to the epidermis or dermal/epidermal junction • Clark level II melanomas invade into the superficial (papillary) dermis, and these are usually radial growth phase lesions. • Clark level III melanomas fill the papillary dermis • Clark level IV melanomas invade into the deep (reticular) dermis and have significant metastatic risk. • Clark level V melanomas are uncommon and contain invasion into the subcutaneous fat.
for thin melanomas, Clark level IV or V is associated with higher risk. • Breslow thickness has an effect on survival, local, regional, and systemic recurrence rates, • Thickness is considered in defining the margins of excision for primary melanomas.
Ulceration • as an important negative prognostic feature • is incorporated in the current staging system • the overall stage assignment groups ulcerated lesions with nonulcerated lesions one T level higher (e.g., T2b and T3a are both stage IIA).
Tumor (T) • TX:Primary tumor cannot be assessed (e.g., shave biopsy or regressed) • T0:No evidence of primary tumor • Tis:Melanoma in situ • T1:≤1 mm in thickness (a. without ulceration or b. with ulceration or level IV or V) • T2:1.01–2 mm in thickness (a. without or b. with ulceration) • T3:2.01–4 mm in thickness (a. without or b. with ulceration) • T4:>4 mm in thickness (a. without or b. with ulceration)
Nodes (N) • N1: 1 lymph node (a. micrometastasisa or b. macrometastasisb) • N2: 2–3 lymph nodes (a. micrometastasisa or b. macrometastasis) c. Satellite or in-transit metastasis without nodal metastasis • N3: ≥4 nodes; matted nodes; or in-transit metastasis or satellites with metastasis in regional node(s)
M1a Skin, subcutaneous tissues, or distant nodes • M1b Lung • M1c All other visceral or any distant metastasis with elevated LDH