Advancing Vaccine Candidate from Preclinical Development to Phase I/IIa Clinical Stage
DESCRIPTION
This document outlines the critical pathway for advancing a vaccine candidate from preclinical development to early clinical stages, specifically Phase I/IIa. It emphasizes the importance of Good Manufacturing Practice (GMP) manufacturing and the necessary preclinical toxicology assessments. This journey is essential for ensuring the safety and efficacy of the vaccine before it is administered to human subjects. Key considerations include regulatory compliance, quality assurance, and thorough testing processes to pave the way for successful vaccine trials.
1 / 1
Télécharger la présentation 

Advancing Vaccine Candidate from Preclinical Development to Phase I/IIa Clinical Stage
An Image/Link below is provided (as is) to download presentation
Download Policy: Content on the Website is provided to you AS IS for your information and personal use and may not be sold / licensed / shared on other websites without getting consent from its author.
Content is provided to you AS IS for your information and personal use only.
Download presentation by click this link.
While downloading, if for some reason you are not able to download a presentation, the publisher may have deleted the file from their server.
During download, if you can't get a presentation, the file might be deleted by the publisher.
E N D
Presentation Transcript
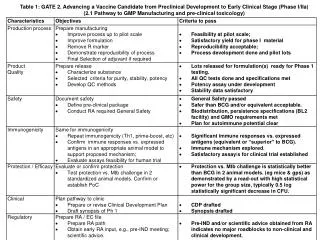
Table 1: GATE 2. Advancing a Vaccine Candidate from Preclinical Development to Early Clinical Stage (Phase I/IIa) • (2.1 Pathway to GMP Manufacturing and pre-clinical toxicology)
More Related