Protein metabolism
Protein metabolism. BY Dr. NAGLAA IBRAHIM AZAB Lecturer Of Medical Biochemistry &Molecular Biology BENHA FACULTY OF MEDICINE. PROTEIN STRUCURE. Protein structure. α- a.a ---- α- a.a ---- α- a.a ---- α- a.a ---------- α- a.a

Protein metabolism
E N D
Presentation Transcript
Protein metabolism BY Dr. NAGLAA IBRAHIM AZAB Lecturer Of Medical Biochemistry &Molecular Biology BENHA FACULTY OF MEDICINE
Protein structure α- a.a ---- α- a.a ---- α- a.a ---- α- a.a ---------- α- a.a 1 2 3 4 n Peptide linkages α- amino acid R—CH—COOH NH2 R1 R2 Rn NH2 -- CH—CO---- NH-- CH—CO----------CO---- NH- CH—COOH Amino terminus peptide linkages carboxyl terminus
Classification of amino acids • • Structural classification : according to the chemical structure of the side chain (R) • • Nutritional classification (essensial & non essential a.as) • • Metabolic classification : according to the fate of amino acids inside the body (glucogenic,ketogenic and mixed a.as)
Aliphatic a.as Neutral a.as Contain One NH2 one COOH Amidic a.as Contain One NH2 one COOH One NH2CO(amidic group) Acidic a.as Contain One NH2 two COOH Basic a.as Contain >One NH2 one COOH
Aliphatic neutral a.as WITH HYDROCARBON SIDE CHAIN -------------------------------------------------- 1-Glycine(2C) CH2–COOH 2-Alanine(3C)CH3–CH–COOH 3-Valine(5C) CH3–CH–CH–COOH 4-Leucine(6C)CH3–CH–CH2–CH–COOH 5-Isoleucine(6C)CH3–CH2–CH–CH–COOH I NH2 I NH2 II CH3NH2 I CH3 I NH2 I CH3 I NH2
WITH HYDROXYL (OH ) CONTAINING SIDE CHAIN ----------------------------------------------------------------------- 1-Serine(3C)CH2–CH–COOH 2-Homoserine(4C)CH2–CH2–CH–COOH 3-Threonine(4C)CH3–CH–CH–COOH I OH I OH I NH2 I OH I NH2 I OH I NH2
WITH SULFUR (S) CONTAINING SIDE CHAIN--------------------------------------------------------------- 1-Cysteine(3C)CH2–CH–COOH 2-Homocysteine(4C)CH2–CH2–CH–COOH 3-Methionine(4C)CH2–CH2–CH–COOH 4-CystineCH2–CH–COOH S S CH2–CH–COOH I SH I NH2 I SH I NH2 I S-CH3 I NH2 I NH2 I NH2
Aliphatic acidic a.as Aspartic acid (4C) HOOC–CH2–CH–COOH Glutamic acid (5C) HOOC–CH2–CH2–CH–COOH Aliphatic Amidic a.as Aspargine (4C) NH2–OC–CH2–CH–COOH Glutamine (5C) NH2–OC–CH2–CH2–CH–COOH I NH2 I NH2 I NH2 I NH2
Aliphatic Basic a.as 1-Lysine (6C)CH2–CH2–CH2–CH2–CH–COOH 2-Arginine (5C+1)CH2–CH2–CH2–CH–COOH 3-Ornithine (5C)CH2–CH2–CH2–CH–COOH 4-Citrulline (5C+1)CH2–CH2–CH2–CH–COOH I NH2 I NH2 NH I NH =C─ NH2 I NH2 Guanido group I NH2 I NH2 I NH2 NH I O= C─ NH2 Ureido group Ornithine and citrulline are not found in proteins but are formed in urea cycle
Aromatic a.as PhenylalanineCH2–CH–COOH Tyrosine = Parahydroxyphenylalanine CH2–CH–COOH I NH2 I NH2 OH
Heterocyclic a.as Tryptophan CH2–CH–COOH Histidine CH2–CH–COOH Proline (imino acid) I NH2 N H I NH2 N NH COOH N H
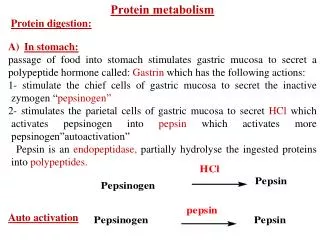
Proteins Parietal cells Gastric HCL . 1st Denatured proteins Chief cells HOW? Endopeptidase with broad Specificity (cleaves the internal peptide bonds in which the carboxylic group is of aromatic a.as or leucine) Pepsinogen Pepsin Inactive zymogen Active enzyme then Rennin in infants Ca Milk caseinogen Soluble casein Ca paracaseinate (milk clot) Denaturation Importance? Large peptides + some free a.as Further digested by pepsin and other proteases
Pancreatic endopeptidasesincluding Intestinal cells Large peptides + some free a.as *Trypsinogen Trypsin (inactive) (active) *Chymotrypsinogen Chymotrypsin (inactive) (active) *Proelastase Elastase (inactive) (active) *Collagenase Cleaves peptide bonds in which the COOH is of Basic a.as (arginine and lysine) Cleaves peptide bonds in which the COOH is of Aromatic a.as or leucine. Cleaves peptide bonds in which the COOH is of small non polar a.as (a.as with small uncharged R as glycine, alanine & serine) Catalyzes the hydrolysis of collagen enteropeptidase Smaller peptides + free a.as
Exopeptidases Pancreatic carboxypeptidasesincluding Cleaves the aromatic a.as from the C- terminal end of the peptide Cleaves the basic a.as from the C- terminal end of the peptide Cleaves one a.a from the N- terminal end of the peptide Cleaves a.as from the dipeptides or tripeptides *Procarboxypeptidase A Carboxy peptidase A (inactive) (active) *Procarboxypeptidase B Carboxy peptidase B (inactive) (active) Smaller peptides + free a.as Trypsin Intestinal proteases *Aminopeptidases *Dipeptidases and tripeptidases a.as
Ininfants, IgAin the clostrum of milk is absorbed without digestion by pinocytosis giving immunity to the babies Intestinal lumen Intestinal cell Blood IgA IgA IgA IgA [gA [gA [gA
Aa.s resulting from protein digestion are absorbed from the small intestine by: ■ passive transport mechanism(For D-aas). ■ Active transport mechanism(For L-aas and dipeptides): ► Carrier protein transport system ( sodium – amino acid carrier system ). ► Glutathione transport system (γ-glutamyl cycle)
Carrier protein transport system ( sodium – amino acid carrier system ) Intestinal lumen K+ Na+ a.a. Na+ a.a. ADP+Pi Cell membrane Na/K ATPase Carrier ATP Intestinal cell K+ Na+ a.a. Cytoplasm Portal blood a.a.
*This system transport the a.a. against its conc. gradiant using energy derived from Na/K+ pump. *Here a.as are absorbed by specific carrier proteinin the cell membrane of the small intestinalcells.This carrier protein has one site for the a.a. and another site for the Na+.It transports them from the intestinal lumen across the cell membrane to the cytoplasm. Then the a.a. passes to the blood down its conc. Gradient, while the Na+ is pumped out from the cell to the intestinal lumen by Na/K+ pump utilizing ATP as a source of energy derived from Na/K+ pump.
Glutathione transport system (γ-glutamyl cycle) R a.a. NH2—CH—COOH γ-glutamyl transpeptidase(transferase) (GGT) Cell membrane CO-NH-CH-COOH CH2 R γ-glutamyl- cysteinylglycine cysteinylglycine CH2 ADP+Pi CH-NH2 glutathione synthetase dipeptidase COOH ATP γ-glutamyla.a. glycine γ-glutamyl- cysteine γ-Glutamyl cyclotransferase a.a. ADP+Pi γ-Glutamylcysteine synthetase cysteine 5-oxoproline ATP H2C CH2 Glutamic acid Oxoprolinase H2O O=C CH-COOH ADP+Pi ATP N I H
►This transport system is for: the transport of a.a.s from the extracellular space to the cytoplasm in the intestine,kidney, brain & liver(bile ductule cells) So it is not important only for the uptake of a.as from the intestinal lumen (a.a. absorption) . ►3 ATP molecules are utilized for the transfer of one a.a. ►Clinical notes: ■The blood conc. of GGT enzyme is increased in cholestasis & chronic alcholism ( so used as a liver function test). ■Oxoprolinuria:Inheriteddeficiency of glutathione synthetase enzyme , leading to increase levels of 5 oxoproline in blood &urine acidosis & neurological damage
Quiz The basis of allergic reactions to food
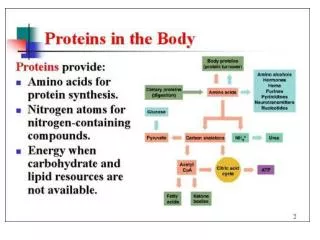
Fate of absorbed a.as Enter in the formation of a.a. pool
►Definition:It include the free a.as distributed throughout the body - The a.a. pool contains 100 gm a.as.50% of these a.as are in the form of glutamate & glutamine (Why?) - In contrast to the amount of protein in the body (about 12 Kg in 70 Kg man), the a.a. pool is small (only 100 gm) ►Sources & fate of the a.a. pool: Non essential a.a.s synthesized in the body Diatery proteins Tissue proteins a.a.s Amino acid pool Anabolism Catabolism(Deamination) Synthesis of Fate of deamination products Proteins Other nitrogenous compounds α- keto acid Ammonia ►Aminosugars ►Nitrogenous bases of phospholipids ►Purines & pyrimidines ►Neurotransmitters ►Niacin ►Creatine ►Heme ►Tissue proteins ►Plasma proteins ►Enzymes ►Some hormones ►Milk Krebs cycle Ketone bodies Synthetic pathway Catabolic pathway glucose CO2+H2O +ENERGY Non essential a.a.synthesis Excreted in urine glutamine Urea
Catabolism of the a.a.s occurs by Deamination (removal of the amino group) either by General methods of deamination: ►Oxidative deamination ►Transamination ►Transdeamination Or Specific methods of deamination: For certain a.a.s
Oxidative deamination ► Definition:It is the oxidation(removal of hydrogen) anddeamination(removal of the amino group which is liberated as free ammonia) giving α- ketoacid and ammonia (reversible reactions) R— C— COOH H NH3 l NH2 l l O
► Site:In most tissues , mostly in the liver and kidney ► Enzymes involved: 1- L-glutamate dehydrogenase enzyme: Present in the cytosol & mitochondria of most tissues HOOC–CH2–CH2–CH–COOH ◄► HOOC–CH2–CH2–C –COOH NAD(P) NAD(P)H+H H2O NH3 L-GLUTAMATE DEHYDROGENASE ENZYME l l O l NH2 L-glutamic acid α- ketoglutaric acid Coenzyme is NAD or NADP
Regulation: The direction of the reaction depends on: 1- Availability of the substrates: --Relative conc. Of (α-ketoglutarate &NH3) and (glutamate). --Ratio of NADP : NADPH+H 2- Allosteric regulation: --Activators : ADP or GDP. -- Inhibitors : ATP ,GTP & NADH
QUIZ ---After a protein meal , in which direction the reaction proceeds? Why? ---After a CHO meal , in which direction the reaction proceeds? Why?
2- D- & L- a.a. oxidases : Present only in the liver and kidney in minimal amounts They are of low activity in the mammalian tissue N.B.a.a.s L-a.as : mammalian proteins are formed of only L-a.as D-a.as:are found in plants and the cell wall of microorganisms but not used in the synthesis of mammalian proteins L-a.a. oxidase:It deaminates most of the naturally occuring a.a.s. R— CH — COOH R— C — COOH NH3 H2O L-a.a. oxidase l NH2 l l O FMN FMNH2 L- amino acid α- keto acid H2O2 O2 H2O2 Catalase 2 H2O + O2
D-a.a. oxidase:It deaminates D-a.as present in diet givingα- keto acids that either transaminated to the coressponding L-a.as or converted to glucose or F.as or catabolized to CO2 + H2O + energy R— CH — COOH R— C — COOH H2O NH3 D-a.a. oxidase l NH2 l l O FAD FADH2 D- amino acid α- keto acid H2O2 O2 L- amino acid Transaminase H2O2 Catalase α- keto acid 2 H2O + O2 ■Glucose ■f.as ■CO2+H2O R— CH — COOH l NH2 L- amino acid
►Importance of oxidative deamination L-glutamate dehydrogenase enzyme is the only a.a. that undergoes oxidative deamination in the mammalian tissue. Oxidative deaminationby L- glutamate dehydrogenase is an essential component oftransdeamination. So it is important in deamination of most a.as.
Transamination ►Definition:It is the transfer of amino group from one α- a.a. to α- keto acid to form anew α- a.a & a new α- keto acid(reversible reaction) ► Enzymes involved: Transaminases or aminotransferases Coenzyme: PLP (Pyridoxal phosphate) R— C H — COOH l NH2 R— C — COOH l l O ► ◄ Transaminase PLP ► ◄ R— C — COOH l l O R— C H — COOH l NH2
►Site:In the cytosol or both the cytosol & the mitochondria of most cells especially the liver ► All a.asexceptthreonine,lysine,proline&hydroxy prolinemay undergo transamination
►α-ketoglutarate & glutamate are often involved in transamination reactions α- a.a. α- keto glutarate ► ◄ Glutamate transaminase ◄ ► α- keto acid Glutamic acid
►Transaminases of clinical importance are: ■ Alanine transaminase(ALT) = { Glutamate pyruvate transaminase(GPT)}: Glutamic acid + Pyruvic acid Alanine + α- keto glutarate ■ Aspartate transaminase(AST)= { Glutamate oxaloacetate transaminase(GOT)}: Glutamic acid + Oxaloacetic acid Aspartic acid + α- keto glutarate ALT PLP CH3— C — COOH l l O CH3— C H — COOH l NH2 ALT PLP HOOC–CH2–CH–COOH l NH2 HOOC–CH2–C–COOH l l O
►Value of transamination: ■Function: 1- Degradation of a.as to form α- keto acids. 2- Synthesis of non essential a.as from CHO. ■Diagnosticvalue: Transaminases are normally intracellular enzymes. They are elevated in the blood when damage to the cells producing these enzymes occurs. ** Increase level of both ALT & AST indicates possible damage to the liver cells. ** Increase level of AST alone suggest damage to heart muscle ,skeletal muscle or kidney.
Transdeamination ►Definition: It is the combination of transamination & oxidative deamination. It includes the transamination of most a.as with α– keto glutarate to form glutamate then the glutamate is oxidatively deaminated reforming α– keto glutarate and giving ammonia. This provides a pathway by which the amino group of most a.as is released in the form of ammonia. ► α- keto glutarate ◄ α- a.a. ◄ ► NH3 ► NAD(P)H+H Glutamate transaminase L-GLUTAMATE DEHYDROGENASE ENZYME NAD(P) ► ► H2O α- keto acid Glutamic acid ◄ ◄
On biochemical basis explain ■Removal of ammonia from a.as can not be explained alone by transamination nor by oxidative deamination alone It can not be explained by transamination alone as no free ammonia is liberated nor by oxidative deamination alone as oxid. Deamination works efficiently only on glutamic acid as L- glutamate dehydrogenase is of high activity in the mammalian tissue,while the L- amino acid oxidase which works on most a.as is of low activity. ■The formation of NH3 from α– a.as occurs mainly via the α– amino nitrogen of glutamate As this occurs by transdeamination, and L- glutamate is the only a.a that undergoes oxidative deamination at an appreciable rate in the mammalian tissue.
Another importance of transdeamination is we can form α– a.a. from ammonia ► α- keto glutarate ◄ α- a.a. ◄ ► NH3 ► NAD(P)H+H Glutamate transaminase L-GLUTAMATE DEHYDROGENASE ENZYME NAD(P) ► ► H2O α- keto acid Glutamic acid ◄ ◄
Specific methods of deamination 1- L- glutamate dehydrogenase: Said before
2- Glycine oxidase: as the mechanism of action of D- amino acid oxidase CH2 — COOH CH — COOH H2O NH3 Glycine oxidase l l O l NH2 FAD FADH2 Glycine Glyoxylic acid H2O2 O2 H2O2 Catalase 2 H2O + O2
3-Glycine cleavage system: + FH4 CH2─FH4 + NH3 + CO2 NAD NADH+H CH2 – COOH | NH2 Tetrahydrofolic acid Methylene tetrahydrofolic acid Glycine
4- Histidase:( Non oxidative deamination) – CH2 – CH – COOH | NH2 Histidase – CH = CH – COOH N NH NH3 N NH Urocanic acid Histidine
5-Dehydratases: (Non oxidative deamination) For hydroxy containing a.as (serine& threonine) CH2–CH–COOH l l OH NH2 CH2=C–COOH l NH2 CH3–C–COOH ll NH Serine dehydratase PLP H2O Serine α–aminoacrylic acid α–iminopropionic acid H2O Serine dehydratase NH3 CH3–C–COOH ll O Pyruvic acid CH3–CH–CH–COOH l l OH NH2 CH3–CH2–C–COOH ll O Threonine dehydratase PLP NH3 Threonine α–ketobutyric acid