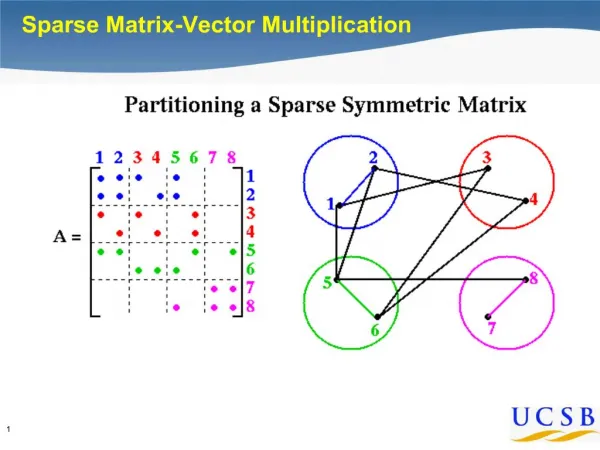
Vector and Scalar Products in Physics
Learn about vector and scalar products, Coulomb's Law, charge densities, conductors vs. insulators, and fundamental concepts in electricity and magnetism. Explore forces and interactions in matter.

Vector and Scalar Products in Physics
E N D
Presentation Transcript
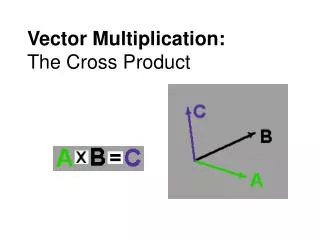
Vector Multiplication There are two ways (in 2 or 3D) to multiply vectors. Scalar product -> two vectors make a scalar A●B =N Also called the dot product or the inner product Vector product -> two vectors make a vector Also called the cross product, alternating product or the outer product A x B = C
Scalar Product Scalar product -> two vectors make a scalar A ●B =ABcos q Geometric A ●B =axbx+ayby+azbz Algebraic
Vector Product Vector product -> two vectors make a vector A x B = C Geometric C has magnitude absinq. Direction perpendicular to the plane containing A and B. A x B = (aybz-byaz)i+(azbx-bzax)j+(axby-bxay)k Algebraic
The right hand rule Force F velocity v Magnetic Field B F=q(v x B)
Electricity and Magnetism • One of the four fundamental forces of nature • Responsible for the vast majority of what we observe around us • Probably best-understood and best-tested of the forces of nature • Electromagnetic Interactions: • Electricity and Electronics • Magnetism • Chemistry • Biology • and even more
Electrical Charges • Electric forces only affect objects with charge • Charge is measured in Coulombs (C). A Coulomb is a large unit of charge. 1 electron has -1.6 x 10-19 C of charge. • Charge comes in both positive and negative quantities • Charge is conserved – it can neither be created nor destroyed • Charge is usually denoted by the letter q. • An object has a total charge of 5 mC. It is divided into two pieces, one of which has charge 8 mC and the other of which has charge • 3 mC • -3 mC • 13 mC • Such a division is impossible
Matter and Charges • All matter is made of positive and negative charges (or neutral) • An object’s total charge is very close to zero • When an object becomes charged, a tiny fraction of its charged particles (usually electrons) are lost or gained • These particles (usually electrons) can flow through objects What do you think of when you hear the words conductor or insulator? Conductor A material that allows electrons or other charged particles to flow freely Insulator A material that resists the flow of electrons and other charged particles
Elementary Charge • Charges exist in integer multiples of a fundamental charge unit called e • We will consider e to be a positive number (some sources treat it as negative) e = 1.602 × 10-19 C the magnitude of the charge on an electron. the charge on a proton When you write that an atom or molecule has a charge +1 , you mean +e. A partial charge means that a charge density can be modeled as having a charge at a location less than e
A cube with side 1 cm has a charge density of = 1 C/m3. What is the charge of the cube? • 1 C • 0.01 C = 10 mC • 10-4 C = 100 m C • 10-6C = 1 mC 1 cm Charge Densities Charge can be localized to discrete points (point charges), or it may be spread out over a volume, a surface or a line • Charge density units C/m3 • Surface charge density units C/m2 • Linear charge density units C/m
Coulomb’s Law • Like charges repel, unlike charges attract • Force is directly along a line joining the two charges q1 q2 r 0= 8.85410-12 C2/ (N●m2) • Permittivity of free space • An inverse square law, just like gravity • Can be attractive or repulsive – unlike gravity • Constant is enormous compared to gravity
ke= 8.988109 Nm2/C2 Coulomb’s Law: Applied A Helium nucleus (charge +2e) is separated from one of its electrons (charge –e) by about 3.00 10-11m. What is the force the nucleus exerts on the electron? Is it attractive or repulsive? r = 3.00 10-11m • We just calculated the force on the electron from the nucleus. How does this compare with the force on the nucleus from the electron? • The force on the nucleus is twice as big • The force on the nucleus is half as big • The forces are equal in magnitude q1 = 3.204 10-19C q2 = -1.602 10-19C • How does the acceleration of the nucleus compare to that of the electron? • The acceleration of the nucleus is larger • The acceleration of the nucleus is smaller • The accelerations are equal. Fe= - 0.513 N Attractive Force
Newton’s Laws and Kinematics Newton’s laws and all the kinematics you learned in 113 are still true! If a does not depend on time, then