Stoichiometry
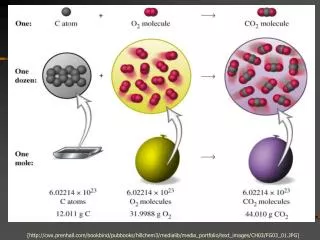
[http://cwx.prenhall.com/bookbind/pubbooks/hillchem3/medialib/media_portfolio/text_images/CH03/FG03_01.JPG]. Stoichiometry. This is the study of quantities, or measurable relationships, that exist in chemical reactions.


Stoichiometry
E N D
Presentation Transcript
[http://cwx.prenhall.com/bookbind/pubbooks/hillchem3/medialib/media_portfolio/text_images/CH03/FG03_01.JPG][http://cwx.prenhall.com/bookbind/pubbooks/hillchem3/medialib/media_portfolio/text_images/CH03/FG03_01.JPG]
Stoichiometry • This is the study of quantities, or measurable relationships, that exist in chemical reactions. • The coefficients in balanced chemical equations always refer to the moles of chemicals that are needed.
Moles to Moles Conversions • Example: • Na + O2 Na2O • If you start with 8.0 moles of Na, how many moles of Na2O will you have when the reaction is complete? 4 2
Moles to Moles Conversions 4 Na + O2 2 Na2O 8.0 mol Na 2 mol Na2O = 4.0 mol Na2O 4 mol Na Significant digits! Mole Ratio from balanced equation. Start with the quantity given to you in the problem.
Mass to Mass Conversions • Example: (Using same reaction…) • If you start with 92.0 g of Na, how many grams of Na2O will you have at the end of the reaction?
Mass to Mass Conversions 4 2 Na + O2 Na2O 92.0 g Na 1 mol Na 2 mol Na2O 62.0 g Na2O 23.0 g Na 4 mol Na 1 mol Na2O = 124 g Na2O
Other Variations…. • Example: • If you start with 54.0 g of Aluminum, how many moles of Oxygen do you need for the reaction to work completely? • Al + O2 Al2O3 4 2 3 2
Other Variations…. Al + O2 Al2O3 4 3 2 54.0 g Al 1 mol Al 3 mol O2 = 1.50 mol O2 27.0 g Al 4 mol Al
Other Variations…. • Example: • If you start with 4.00 moles of Oxygen, how many molecules of Magnesium Oxide will you have at the end of the reaction? • Mg + O2 MgO 2 2
Other Variations…. Mg + O2 MgO 2 2 2 mol MgO 4.00 mol O2 6.02 x 1023 molec MgO 1 mol O2 1 mol MgO = 4.82 x 1024 molec MgO