ENZYME CATALYSIS LAB
ENZYME CATALYSIS LAB. AP Lab #2 AP Biology. FIRST…. Let’s review enzymes!. RECALL …Enzymes are:. A specific type of protein Speeds up (catalyze) chemical reactions by lowering activation E Are NOT used up during rxns instead are recycled/reused

ENZYME CATALYSIS LAB
E N D
Presentation Transcript
ENZYME CATALYSIS LAB AP Lab #2 AP Biology
FIRST…. Let’s review enzymes!
RECALL…Enzymes are: • A specific type of protein • Speeds up (catalyze) chemical reactions by lowering activation E • Are NOT used up during rxns instead are recycled/reused • Have primary, secondary, teritary, and (sometimes) quartnary structures • Can be denatured (just like any protein) de= take away; nature = function
http://www.stolaf.edu/people/giannini/flashanimat/enzymes/transition%20state.swfhttp://www.stolaf.edu/people/giannini/flashanimat/enzymes/transition%20state.swf
http://www.stolaf.edu/people/giannini/flashanimat/proteins/protein%20structure.swfhttp://www.stolaf.edu/people/giannini/flashanimat/proteins/protein%20structure.swf
WHAT DO THE “R GROUPS” DO IN PROTEINS? • Each amino acid (monomer of proteins) has UNIQUE combo of R-groups attached to their carbon skeleton • Involved in secondary/tertiary structure bonding • Nonpolar Amino Acids: • Gly, Ala, Val, Leu, Ile, Met, Phe, Trp, Pro • the charges are spread across the molecule • Polar Amino Acids: • Ser, Thr, Cys, Tyr, Asn, Gln- • have opposite charges on opposite sides
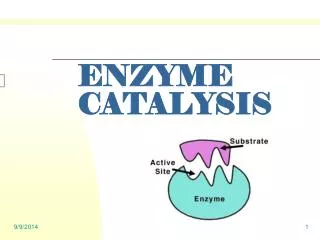
Binding Specificity • Even when different substrate molecules are present, only those that have the specific shape complementary to the active site are able to bind with the enzyme's active site.
INDUCE FIT • Once substrate binds… • Active site undergoes conformational change (changes shape) • enhances catalysis speeds up substrate to product conversion b/c allows bonds to break/be made more quickly • http://www.stolaf.edu/people/giannini/flashanimat/enzymes/enzyme.swf
ENZYMES ARE RECYCLED • Release of products restores enzyme to its ORIGINAL conformation (shape) • Enzyme can repeat this reaction over and over, as long as substrate molecules are present
ENZYME CONFORMATION (SHAPE) • maintained by interactions btw R groups • sensitive to changes in enzyme's environment • 2 important influences are: • pH and temperature • When enzyme's conformation is significantly altered because of pH or temperature variation denaturation occurs enzyme may no longer catalyze reactions • An enzyme is said to be denatured when it loses its functional shape.
WHAT FACTORS DENATURE PROTEINS? • pH • Salt concentration • Temperature
WAIT…WHAT IS “DENATURATION” AGAIN? • the process through which the protein unravels and loses its native conformation/shape (3D shape (teritary) and secondary structures are disrupted!) • Enzyme function is lowered or stops
WHY DOES PH DENATURE PROTEINS? • In either excess H+ or excess OH- ions, protein's shape is altered • Active site is distorted/blocked alters ionic bonds that contribute to its functional shape • Enzyme cannot catalyze reactions at all or as well • http://www.lewport.wnyric.org/JWANAMAKER/animations/Enzyme%20activity.html
WHY DOES SALT [ ] DENATURE PROTEINS? • In either excess + or excess - ions, protein's shape is altered • R-groups/side chains of amino acids distorted/blocked by affecting ionic bonding • Enzyme cannot catalyze reactions at b/c it forms a precipice • Less salt= R groups form extra bonds with each other • More salt= disrupt R groups from normal bonding patterns
WHY DOES TEMPERATUREDENATURE PROTEINS? • Kinetic energy changes • Atoms move differently and affects the bonding patterns that hold the protein together • A higher temperature generally results in increase activity b/c molecular motion increases resulting in more molecular collisions • If, however, temp rises above a certain point, the heat will denature molecules move too fast and can’t H-bond • Cold temp’s SLOW DOWN or stop activityb/c molecular motion decrease • http://www.lewport.wnyric.org/JWANAMAKER/animations/Enzyme%20activity.html
[SUBSTRATE] ALSO EFFECTS ENZYME ACTIVITY • If [ ] of enzyme is constant… • at lower [substrate] [substrate]= limiting factor • As [substrate] increases, RATE of enzyme activity also increases • However, at very high [substrate] enzymes become saturated with substrate and a higher concentration of substrate does NOTHING to increase the reaction rate • All the enzymes are already in use
INTRODUCTION TO … Lab #2: Enzyme Catalysis
CONCEPTS • Understand substrates and products, enzyme structure & denaturation • Measure factors affecting enzyme activity • Measure rate ofO2 production • Experimental design • rate of reactivity • reaction with enzyme vs. reaction without enzyme • optimum pH or temperature • test at various pH or temperature values
KEY CONCEPTS • What is are the reactants(substrate) & products? • What enzyme are we using? • H2O2 H2O + O2 Hydrogen water + oxygen gas peroxide catalase
NOTE: Oxygen gas(O2) forms BUBBLES ENZYME SUBSTRATE (REACTANT) PRODUCTS
WHY DOES H2O2 NEED TO BE BROKEN DOWN? • H2O2 is poisonous (TOXIC) to cells; water and O2 are NOT • H2O2 is naturally produced as a byproduct of oxygen metabolism (cellular respiration) • Almost all organisms possess enzymes known collectively as PEROXIDASES (ex: catalase) found in PEROXISOMES. • Converts H2O2 into harmless H2Oand O2
The nature and arrangement of amino acids in the active site make it specific for only one type of substrate. The nature and arrangement of amino acids in the active site make it specific for only one type of substrate. NOTE: The nature and arrangement of amino acids in the active site make it specific for only one type of substrate.
PROCEDURE (USE WARD’S) • PART A: Testing an Enzyme • THIS WILL BE A DEMO BY TEACHER!! • PART B: Establishing a BASELINE by determining the amount of H2O2 in 1.5% solution • PART C: Determining the Rate of H2O2 Spontaneous Decomposition • PART D: Determining the Rate of H2O2 Decomposition by Enzyme Catalysis
Reading the titration syringe • Remember to read INITIAL and FINAL • Use MENISCUS!
TITRATION RXN • The sulfuric acid (H2SO4) will “freeze” enzyme reaction. WHY??? • Adding H+ ions disrupts active site • The Titration Reaction is below: 5 H2O2 + 2 KMNO4 + 3 H2SO4 K2SO4 + 2 MnSO4 + 8 H2O + 5 O2 ** H2O2 reacts with 2 KMNO4 ; once H2O2 all used up KMNO4 can’t react anymore HINT: TITRATION SHOULD STAY A FAINT PINK COLOR…Brown = too much titrant!!!
MORE LAB HINTS • Titrate only 5 ml of the sample at a time. • This way, if you exceed the endpoint or have an error in titration, you will have sufficient sample to repeat the titration. • CONTROL the sample same size they need to be ALL the SAME (5 ml) • Place solution to be titrated over a piece of WHITE paper so you can see color changes easily. • Swirl (do not shake) sample after every few drops to mix well • For each sample, be sure to stop the titration at the same color
ANALYSIS OF DATA Calculate the RATE OF ENZYMATIC REACTIUON
CONCLUSIONS • Enzyme reaction rate is affected by: • pH, temp, [substrate], & [enzyme]
SOME REAL-LIFE APPLICATIONS…. • Bacterial enzymes and use of disinfectants • Many disinfectants, such as chlorine, iodine, iodophores, mercurials, silver nitrate, formaldehyde, and ethylene oxide, INACTIVATE bacterial enzymes and block metabolism. • Extremes of temperature to control bacteria. • High temperatures, such as autoclaving, boiling, and pasteurization, denature proteins and STOP functions • Cold temperatures, such as refrigeration or freezing, SLOW DOWN or STOP enzyme rxns
LET’S REVIEW THE LAB…. • Part A: Demo Testing Catalase Activity in Liver • Purpose = ? • To determine the effect of temperature on catalase activity • Hypotheses = ? • If catalase is present then oxygen gas will be formed because catalase will break down its substrate, hydrogen peroxide, to make its product, O2. • If the temperature is very high around catalase then oxygen gas will not form because catalase will become denatured.
LET’S REVIEW THE LAB…. • Part A: Demo Testing Catalysis Activity in Liver • CONTROL = ? • Catalase/Liver at Room Temp • Independent variable (what are you testing/changing?) =? • Temperature • Dependent variable (what are you measuring?) =? • Amount of oxygen gas (NOT # of Bubbles)
LET’S REVIEW THE LAB…. • Part C: Rate of H2O2 Spontaneous Decomposition • Purpose = ? • To determine if the breakdown of H2O2 is a spontaneous, exergonic reaction. • Hypothesis = ? • If H2O2 is an exergonic reaction then there will be less H2O2 after 120 hours (or 5 days) because breaks down spontaneously.
LET’S REVIEW THE LAB…. • Part C: Rate of H2O2 Spontaneous Decomposition • CONTROL = ? • Baseline (amount of H2O2 in original 1.5% H2O2 solution used) • taken in Part B • Independent variable (what are you testing/changing?) =? • Time (hours) • Dependent variable (what are you measuring?) =? • Amount of H2O2 decomposition (NOT # of KMnO4)
LET’S REVIEW THE LAB…. • Part D: Rate of H2O2 Decomposition Using Catalase Enzyme • Purpose = ? • To determine the rate of H2O2 decomposition OR… • To determine the rate of enzymatic reaction • Hypothesis = ? • If catalase is functioning longer and not denatured by acid then there will be more H2O2 decomposition because catalase will break down more H2O2.
LET’S REVIEW THE LAB…. • Part D: Rate of H2O2 Decomposition Using Catalase Enzyme • CONTROL = ? • Baseline (amount of H2O2 in original 1.5% H2O2 solution used) • taken in Part B • Independent variable (what are you testing/changing?) =? • Time catalase is working (seconds) • Dependent variable (what are you measuring?) =? • Amount of H2O2 decomposition (NOT # of KMnO4)