Robert Millikan
Robert Millikan. By: Brigitte Jacoby and Steph Wall. Robert Millikan's Background. He was born on March 22, 1868 in Illinois He died on December 19, 1953 Robert Millikan had 5 other brothers and sisters He went to Oberlin College in Ohio, as did both of his parents

Robert Millikan
E N D
Presentation Transcript
Robert Millikan By: Brigitte Jacoby and Steph Wall
Robert Millikan's Background He was born on March 22, 1868 in Illinois He died on December 19, 1953 Robert Millikan had 5 other brothers and sisters He went to Oberlin College in Ohio, as did both of his parents He was asked to teach a basic physics course for a semester at Oberlin College Robert became interested in physics when preparing for this task He was the only physics graduate of his class Millikan married Greta Irvin Blanchard in 1902 and they had three sons He retired in 1946 and his autobiography was published in 1950
Millikan’s Scientific Research Robert was fascinated by electricity and found the charge of the electron He helped create anti-submarine devices during WWI as Vice Chairman of the National Research Council Millikan, in 1921, became part of the California Institute of Technology He was involved in studies of radiation that Victor Hess had detected impacting Earth from outer space (now called cosmic rays)
Science Achievement Millikan proved that the electron was a fundamental particle with a fundamental charge in his famous oil-drop experiment In 1913, he published his value for the charge, which remained the accepted value for decades The determination forvalue of the electron, and the ingenuity of his oil-dropexperiment, earned Millikan international recognition and the 1923 Nobel Prize in physics. He published 6 physics textbooks and served as president in the American Physical Society Millikan was also VP of the American Association for the Advancement of Science
Why Millikan Decided to Preform the Oil-Drop Experiment H.A. Wilson first developed the standard technique for measuring charge This process used water, not oil Water droplets would be given an electric charge and the rate at which they fell would be measured An electric field attracted the charged droplets upward and opposed the force of gravity By determining the strength of the electric field and the mass of the water droplets, he could calculate the charge on the droplets and on the electrons. Millikan tried Wilson's technique but found that the rapid evaporation of the water droplets made measuring difficult and the results unpredictable
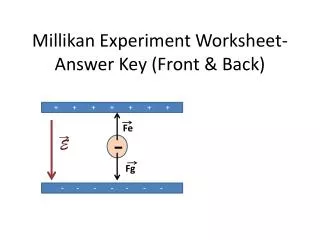
Oil-Drop Experiment Using an atomizer Millikan thinly sprayed oil droplets above two parallel plates (an atomizer is a device that turns a stream of liquid into a spray) The droplets were falling through a small hole in the upper plate to the space between them The droplets could be seen at one side through a short focal distance telescope To make these droplets more visible Robert lit them up from one side The view was intersected by two very small, horizontal lines. Droplets of oil could electrify themselves by owing to friction when they were atomized
Oil-Drop Experiment cont. The oil droplets could also get a charge from an X-ray to complete a potential electric field By doing this, he discovered how the charge of the droplets affected the way they fell In the end, after many trials of his experiments, Robert Millikan discovered the charge of the electron was negative
Citations "Robert A. Millikan." Notable Scientists from 1900 to the Present. Ed. Brigham Narins. Detroit: Gale Group, 2008. Gale Science In Context. Web. 7 Oct. 2011. "Robert Andrews Millikan." Science and Its Times. Ed. Neil Schlager and Josh Lauer. Vol. 6. Detroit: Gale, 2000. Gale Science In Context. Web. 7 Oct. 2011. Millikan's experiment. (n.d.). Retrieved October 10, 2011, from ThinkQuest website: http://library.thinkquest.org/19662/low/eng/exp-millikan.html American Physical Society to commemorate University of Chicago as historic physics site in honor of Nobel laureate Robert Millikan. (2006, November 28). Retrieved October 14, 2011, from http://www-news.uchicago.edu/releases/ 06/061128.millikan.shtml