Chemical Bonding: Types, Structures & Applications
190 likes | 214 Vues
Explore why atoms stick together through ionic, covalent, and metallic bonding. Learn about valence electrons, Lewis structures, and molecular properties. Practice drawing structures and understanding bond reactivity.

Chemical Bonding: Types, Structures & Applications
E N D
Presentation Transcript
Chemical Bonding Why do atoms stick together, anyway? Who cares?
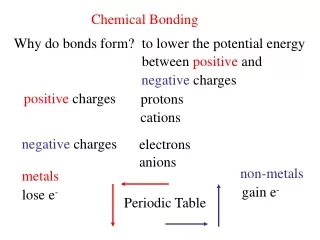
Types of Bonding • Ionic • Covalent • Metallic Ionic Bonding: Covalent Bonding:
How many valence electrons does a Si atom have? • 1 • 2 • 3 • 4 • 5 • 6 • 7 • 8
Lewis Electron-Dot Structures:Describing Bonding in p-block compounds
How many total bonding electrons does an acetate ion have? 1. 3.5 • 7 • 8 • 10 • 12 • 13 • 14 • 15
Why bother? Resonance Number of Bonds Molecular Structure Reactivity Lewis Structure Polarity Physical State and Properties Spectroscopy Hydrophilicity and phobicity
Back to Drawing Lewis Structures I have an idea. Do what I say.
Examples: Reading Organic Structures CH3CH3 CH3CN CHCCHCH2
Draw the Lewis structure for SO3. How many lone pairs does it have? • 1 • 2 • 3 • 4 • 0
Draw the Lewis structure for NO2-. How many lone pairs does it have? • 1 • 2 • 3 • 4 • 0