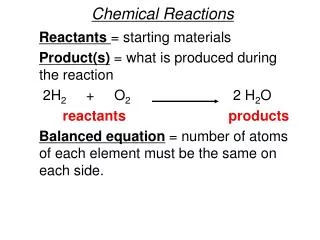
Chemical Reactions
Chemical Reactions. Quick Review. Matter is the “stuff” that makes up the universe. mass. volume. All matter has ______ and ________. All matter is composed of basic building blocks known as _______. atoms. Quick Review.


Chemical Reactions
E N D
Presentation Transcript
Quick Review Matter is the “stuff” that makes up the universe. mass volume All matter has ______ and ________. All matter is composed of basic building blocks known as _______. atoms
Quick Review We know that matter can undergo changes both _________ and __________. physical chemical
Quick Review When matter undergoes a physical change the composition of the substance is unchanged. When matter undergoes a chemical change or reaction the composition of the substance is changed and chemical bonds are formed.
What happens during a chemical reaction? • Atoms in the reactants are rearranged to form one or more different substances (products). • Old bonds are broken; new bonds form. ReactantsProducts MgO + C CO + Mg
How do you know a chemical reaction has occurred? • Evolution of heat and light • Formation of a gas • Formation of a precipitate (formation of a solid in a solution) • Color change • Temperature changes (endothermic/exothermic) • A new odor is produced
Learning Check 1 Classify each of the following as a 1) physical change or 2) chemical change A. ____ a burning candle B. ____ melting ice C. ____ toasting a marshmallow D. ____ cutting a pizza E. ____ polishing silver
Solution 1 Classify each of the following as a 1) physical change or 2) chemical change A. __2__ a burning candle B. __1_ melting ice C. __2__ toasting a marshmallow D. __1__ cutting a pizza E. __2__ polishing silver
A Chemical Reaction Reactants Products
Learning Check 2 A. How does an equation indicate a change in the identity of the reacting substances? B. How did the black and white reactants combine? C. Did all the reactants form product? Why or why not?
Solution 2 A.How does an equation indicate a change in the identity of the reacting substances? The formulas of the reactants are different than the formulas of the products. B. How did the black and white reactants combine? 1 black combined with 1 white. C. Did all the reactants form product? Why or why not? No. There were more black reactants than white.
Mind Jogger • When an object burns, the quantity of ashes that remain is smaller than the original object that was burned. How do you account for the change in mass? Where is the matter that appears to be lost?
Law of Conservation of Mass • mass is neither created nor destroyed in a chemical reaction • total mass stays the same (mass of reactants = mass of products) • atoms can only rearrange 4 H 2 O 4 H 2 O 36 g 4 g 32 g
Law of Conservation of Mass • http://web.visionlearning.com/dalton_playhouse/ad_loader.html
Analyze the Problem The law of conservation of mass will allow us to determine the amount of a missing reactant or product that was involved in a chemical reaction. For example, if you were told that 4 grams of hydrogen reacted with some oxygen to make 36 grams of water then you could figure out how much oxygen was used.
Learning Check 3 • In the complete reaction of 22.99 g of sodium with 35.45 g of chlorine, what mass of sodium chloride is formed? • A 10.0 g sample of magnesium reacts with oxygen to form 16.6 g of magnesium oxide. How many grams of oxygen reacted?
Solution 3 • In the complete reaction of 22.99 g of sodium with 35.45 g of chlorine, what mass of sodium chloride is formed? 81.43 g of Sodium chloride • A 10.0 g sample of magnesium reacts with oxygen to form 16.6 g of magnesium oxide. How many grams of oxygen reacted? 6.6 g of Oxygen
How do we represent these chemical reactions? • Chemists use statements called equations. • Chemical equations do not express numerical equalities as in math. • Chemical equations show the direction in which the reaction progresses. Instead of an equal sign (=), an arrow is used (means yield)
Word Equations • A statement using words to describe a chemical reaction. Sodium (s) + Chlorine (g) Sodium Chloride (s) The statement reads sodium and chlorine react to produce or yield sodium chloride. • Useful but lacks important information
Skeletal Equation • Uses chemical formulas instead of words to identify reactants and products. • Important step in writing a complete equation Na(s) + Cl2(g) NaCl(s)
Learning Check 4 • Write skeleton equations for the following word equations. hydrogen(g) + bromine(g) hydrogen bromide(g) carbon(s) + sulfur(s) carbon disulfide(s)
Solution 4 • Write skeleton equations for the following word equations. hydrogen(g) + bromine(g) hydrogen bromide(g) H2(g) + Br2(g) HBr(g) carbon(s) + sulfur(s) carbon disulfide(s) C(s) + S(s) CS2(s)
Writing a Chemical Equation Chemical symbols give a “before-and-after” picture of a chemical reaction ReactantsProducts MgO + C CO + Mg magnesium oxide to formcarbon monoxide reacts with carbon and magnesium
Balancing a Chemical Equation Balanced means that “conservation of mass” is upheld. Same numbers of each type of atom on each side of the equation Al + S Al2S3 Not Balanced 2Al + 3S Al2S3Balanced
Matter Is Conserved H2 + Cl2 2 HCl + + Total atoms=Total atoms 2 H, 2 Cl 2H, 2 Cl Total Mass=Total Mass 2(1.0) + 2(35.5) 2(36.5) 73.0 g = 73.0 g
Balance Equations with Coefficients Use a number before the compound/element symbol to indicate how many of them are needed called a COEFFICIENT. Coefficients in front of formulas balance each type of atom. 4NH3 + 5O24NO +6H2O 4 N = 4 N 12 H = 12 H 10 O = 10 O
2. Count Number of atoms for each element on both sides How to write and balance Chemical Equations • Write the skeleton equation: Al + O2→ Al2O3 This is not balanced because the numbers don’t match 3. Multiply coefficients until they match – multiply the entire units 2 Al + O2 → Al2O3 Go to 6 oxygens
Al + 2 Al2O3 O2 Balancing Equations Al + O2 2 Al2O3 Multiply each atom by 2 4 3 Balanced
Learning Check 5 Balance each equation. A. Mg + N2 Mg3N2 B. Al + Cl2 AlCl3
Solution 5 A. 3 Mg + N2 Mg3N2 B. 2 Al + 3 Cl2 2 AlCl3
What not to do when balancing Chemical Equations • Neverchange a subscript to balance an equation (You can only change coefficients) • If you change the subscript (formula) you are describing a different chemical. • H2O is a different compound than H2O2 • Neverput a coefficient in the middle of a formula; they must go only in the front. 2NaCl is okay, but Na2Cl is not.