Electrochemistry
Electrochemistry. The electricity produced by chemical reactions or ….. The chemical changes brought about by electricity. Electrochemical reactions = Oxidation-Reduction Reactions. Cell = System where chemical reactions occur. Electrode = means of adding/removing electric current

Electrochemistry
E N D
Presentation Transcript
Electrochemistry The electricity produced by chemical reactions or ….. The chemical changes brought about by electricity. Electrochemical reactions = Oxidation-Reduction Reactions Cell = System where chemical reactions occur Electrode = means of adding/removing electric current to/from system. Cathode = electrode at which reduction occurs. (usually -) Anode = electrode at which oxidation occurs. (usually +) Conductivity = flow of electrons many solid metals conduct electricity electrolyte solutions (acids, bases, or soluble ionic cpds)
Electrochemistry – Cell Types Electrolytic Cells – External source of electricity drives a nonspontaneous (DG > 0) reaction. Electrolysis Used to convert water into H2 and O2 gas. Used for electroplating – put a thin layer of one metal unto another metal at cathode. Voltaic Cells – A spontaneous (DG < 0) reaction is used to generate an electric current. e.g. batteries
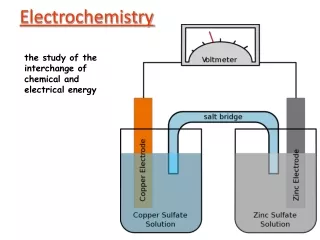
1.100 V e- e- + Zn Cu Salt bridge (KCl – 5% agar) Cl- K+ Zn2+ Cu2+ 1M ZnSO4 Zn(s) Zn2+ + 2e- Oxidation - anode Cu2+ + 2e- Cu(s) reduction - cathode 1M CuSO4 Voltaic Cell Zn Cu Zn|Zn2+(1M)||Cu2+(1M)|Cu
0.462 V e- e- + Cu Ag Salt bridge (KCl – 5% agar) Cl- K+ Cu2+ Ag+ 1M CuSO4 Cu(s) Cu2+ + 2e- Oxidation - anode Ag+ + e- Ag(s) reduction - cathode 1M AgNO3 Voltaic Cell Cu|Cu2+(1M)||Ag+(1M)|Ag
0.763 V e- e- + Zn H2 Salt bridge (KCl – 5% agar) E (SHE) = 0 Cl- K+ E = -0.763 Zn2+ H+ 1M ZnCl2 Zn(s) Zn2+ + 2e- Oxidation - anode 2H+ + 2e- H2(g) reduction - cathode 1M HCl SHE Standard Hydrogen electrode Zn|Zn2+(1M)||H+(1M);H2(1atm)|Pt Zn
0.337 V e- e- + Cu Salt bridge (KCl – 5% agar) E (SHE) = 0 Cl- K+ E = 0.337 H+ Cu2+ 1M HCl(aq) H2(g) 2H+ + 2e- Oxidation - anode Cu2+ + 2e- Cu(s) reduction - cathode 1M CuSO4 Voltaic Cell Pt|H2(1atm);H+(1M)||Cu2+(1M)|Cu H2 Cu
Reduction ½ rx Standard Reduction Potential E volts Zn2+ + 2e- Zn(s) -0.763 V Fe2+ + 2e- Fe(s) -0.44 V Ni2+ + 2e- Ni(s) -0.25 V Pb2+ + 2e- Pb(s) -0.126 V 2H+ + 2e- H2(g) 0.000 V Cu2+ + 2e- Cu(s) +0.337 V Hg2+ + 2e- Hg(s) +0.789 V Ag2+ + e- Ag(s) +0.799 V Cl2 + 2e- 2Cl- +1.360 V The higher up a ½ rx is on the table the more readily that element/substance is oxidized. (Reverse ½ reaction & E˚)
Calculating The Standard Electrical Potential for any cell from the tabulated Standard Reduction Potentials. 1. Choose the appropriate ½ rxs from table 2. Write the ½ rx for the more (+) or less (-) substance • Write the ½ rx for the less (+) or more (-) substance • as an oxidation reaction. (reverse the sign on E) • Write the net balanced reaction (the electrons must be balanced • but do not multiply E by the balancing coefficient!) • Sum the values of E for each ½ rx to get the Cell’s • standard electrical potential.
Reduction ½ rx Standard Reduction Potential E volts Zn2+ + 2e- Zn(s) -0.763 V Fe2+ + 2e- Fe(s) -0.44 V Ni2+ + 2e- Ni(s) -0.25 V Pb2+ + 2e- Pb(s) -0.126 V 2H+ + 2e- H2(g) 0.000 V Cu2+ + 2e- Cu(s) +0.337 V Hg2+ + 2e- Hg(s) +0.789 V Ag2+ + e- Ag(s) +0.799 V Cl2 + 2e- 2Cl- +1.360 V The higher up a ½ rx is on the table the more readily that element/substance is oxidized. (Reverse ½ reaction & E˚)
Reaction Spontaneity and DG DG < 0 reaction is spontaneous – proceeds as written DG = 0 reaction is at equilibrium DG > 0 reaction proceeds in the reverse direction unless enough energy provided to drive reaction forward. DG = DG + RT ln Q DG = - RT ln K • Represents standard conditions where P = 1atm and the [ ]s of all reagents are 1M. Q = 1
Reaction Spontaneity and E E > 0 reaction is spontaneous – proceeds as written with voltage output E = 0 reaction is at equilibrium – no current flow E < 0 reaction proceeds in the reverse direction unless enough current provided to drive reaction forward. (e.g. electrolysis) The Nernst Equation E = E - 2.303RT log Q nF DG = DG + RT ln Q
??? V e- e- + Zn Cu Salt bridge (KCl – 5% agar) Cl- K+ Zn2+ Cu2+ 1.2 M ZnSO4 Zn(s) Zn2+ + 2e- Oxidation - anode Cu2+ + 2e- Cu(s) reduction - cathode 0.80 M CuSO4 Voltaic Cell Zn Cu Zn|Zn2+(1M)||Cu2+(1M)|Cu E = -0.763 - 0.0592 log(1/1.2) n
The Nernst Equation E = E - 2.303 RT log Q lump constants together…. nF E = E - 0.0592 log Q at 25 C n E = 0.0592 log K = RT ln K at 25 C n nF Zn|Zn2+(1M)||Cu2+(1M)|Cu E = 1.10 V Zn|Zn2+(1.2M)||Cu2+(0.8M)|Cu E = ??? V
96,485 C (J V-1 mol-1) Reaction Spontaneity and the Nernst Equation E = E - 0.0592 log Q at 25 C n E = 0.0592 log K = RT ln K at 25 C n nF DG˚ = -RT ln K DG˚ = -nFE˚ or DG = -nFE Calculate DG˚ or DG and K from cell potentials, E˚/E.
anode cathode Electrolysis of Water 2H2O2H2(g) + O2(g) Reduction: (2H+ + 2e- H2(g)) 2 Oxidation: 2H2O 4H+ + O2 + 4e-
Na+ Cl- Na+ + e- Na (floats) Reduction - cathode 2Cl- Cl2(g) + 2e- oxidation - anode Graphite or Platinum are common inert electrodes + Molten NaCl (801˚ C) Electrolysis of molten NaCl
Electron Stoichiometry A Coulomb (C) is the SI unit of charge 1 e- = 1.602 x 10-19 C or 1.602 x 10-19 C per e- 1 mole of e- = 96,485 C = 1 Faraday (F) Current = charge per time = C s-1 = Ampere (A) How many grams of H2 gas can be produced from water through which 1.35 x 106 C have been passed? Reduction: 2H+ + 2e- H2(g)
3.4 amps for 15 seconds – How much Cu is electroplated? Cu(s)→ Cu2+ + 2e- oxidation – anode electrode ‘dissolves’ Cu2+ + 2e- → Cu(s) reduction – cathode Cu plated onto electrode + Cu2+ SO42- Electroplating copper 3.4C x 15s x 1 mol e- x 1 mol Cu x 63.546g Cu = 16.8 mg 1s 96,485C 2 mol e- 1 mol Cu
Anode = Zn Cathode = C 1.6 V Dry Cell ZnCl2; NH4Cl; MnO2 paste Zn + 2NH4+→ Zn2+ + 2NH3 + H2 Anode: (oxidation) Zn → Zn2+ + 2e- Cathode: (reduction) 2NH4+ + 2e-→ 2NH3 + H2 2MnO2 + H2→ 2MnO(OH) (removes H2 gas) Zn2+ + 4NH3 → [Zn (NH3)4]2+ (removes NH3)
Anode = Zn Cathode = C 1.5 V Alkaline Cell ZnCl2; KOH; MnO2 paste Zn + 2MnO2 + 2H2O → Zn(OH)2(s) + 2MnO(OH) Anode: (oxidation) Zn + 2OH-→ Zn(OH)2 + 2e- Cathode: (reduction) 2MnO2 + 2H2O+ 2e-→ 2MnO(OH) + 2OH- Alkaline cells have longer shelf lives
Anode = Cd Cathode = NiO2 1.4 V Ni - Cd rechargeable Ni(OH)2(s); KOH; Cd(OH)2(s) Cd + NiO2(s) + 2H2O → Cd(OH)2(s) + Ni(OH)2(s) Rechargeable Batteries – Nickel-Cadmium Anode: (oxidation) Cd + 2OH-→ Cd(OH)2(s) + 2e- Cathode: (reduction) NiO2 + 2H2O+ 2e-→ Ni(OH)2(s) + 2OH-