No Significant Difference in Clinical Outcomes of Carvedilol Doses in Heart Failure Patients
This study evaluates the clinical outcomes of patients with chronic stable heart failure receiving carvedilol at varying daily doses (2.5 mg, 5 mg, and 20 mg) over a mean follow-up of 3 years. The findings indicate no significant differences in death or hospitalization rates among the groups, with all-cause mortality and hospitalizations for cardiovascular diseases remaining statistically non-significant. The study highlights the need for further research due to its underpowered design to detect differences in important clinical outcomes.

No Significant Difference in Clinical Outcomes of Carvedilol Doses in Heart Failure Patients
E N D
Presentation Transcript
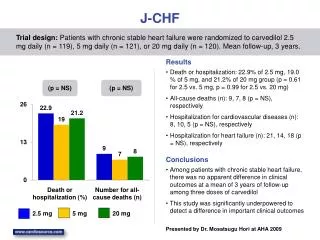
J-CHF Death or hospitalization: 22.9% of 2.5 mg, 19.0 % of 5 mg, and 21.2% of 20 mg group (p = 0.61 for 2.5 vs. 5 mg, p = 0.99 for 2.5 vs. 20 mg) All-cause deaths (n): 9, 7, 8 (p = NS), respectively Hospitalization for cardiovascular diseases (n): 8, 10, 5 (p = NS), respectively Hospitalization for heart failure (n): 21, 14, 18 (p = NS), respectively Trial design: Patients with chronic stable heart failure were randomized to carvedilol 2.5 mg daily (n = 119), 5 mg daily (n = 121), or 20 mg daily (n = 120). Mean follow-up, 3 years. Results (p = NS) (p = NS) Conclusions • Among patients with chronic stable heart failure, there was no apparent difference in clinical outcomes at a mean of 3 years of follow-up among three doses of carvedilol • This study was significantly underpowered to detect a difference in important clinical outcomes Death or hospitalization (%) Number for all-cause deaths (n) 2.5 mg 5 mg 20 mg Presented by Dr. Mosatsugu Hori at AHA 2009