Understanding Molecular Orbital Theory: Interactions of Atomic Orbitals in Small Molecules
This chapter explores the interactions of various atomic orbitals, focusing on homonuclear diatomic molecules such as H2. It discusses combinations of 1s wavefunctions, interactions of s, p, and d orbitals, and how these interactions affect energy levels. The chapter also covers molecular orbital (MO) diagrams, examples in water (H2O), methane (CH4), and the application of symmetry and Linear Combination of Atomic Orbitals (LCAO). Evidence from photoelectron spectroscopy helps understand relative atomic orbital energies and hybrid orbitals.

Understanding Molecular Orbital Theory: Interactions of Atomic Orbitals in Small Molecules
E N D
Presentation Transcript
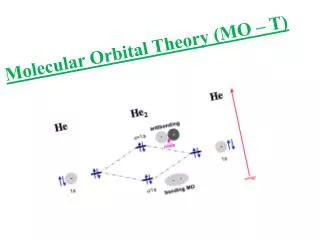
Interactions of two s orbitals: H2 Look at combinations of 1s wavefunctions:
Orbital Mixing Orbitals of samesymmetry interact.lower E orbital decreasesin E.higher e orbital increases in E.
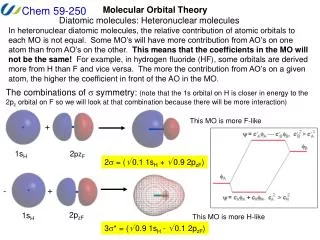
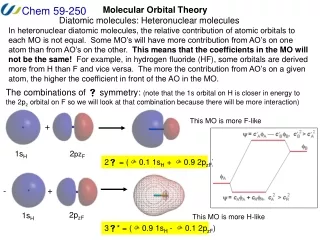
What about different atoms? How do you know relative atomic orbital energies?
More complicated: Using symmetry and LCAO’s H2O
More complicated: Using symmetry and LCAO’s H2O
More complicated: Using symmetry and LCAO’s H2O
CH4Mos: LCAO’s for H’s in Td point group. Then relate those to C’s orbitals to predict MOs.