Understanding pH: The Scale of Acidity and Alkalinity
pH measures the concentration of hydrogen ions (H+) in a solution, determining its acidity or alkalinity. A scale ranging from 0 to 14 is used, where pH < 7 indicates acidity (higher H+ concentration) and pH > 7 indicates alkalinity (higher OH- concentration). Key properties of acids include a sour taste and the ability to turn blue litmus paper red, while bases are slippery, bitter, and turn red litmus paper blue. Each unit decrease in pH represents a tenfold increase in H+ concentration, highlighting the importance of understanding and measuring pH in various solutions.

Understanding pH: The Scale of Acidity and Alkalinity
E N D
Presentation Transcript
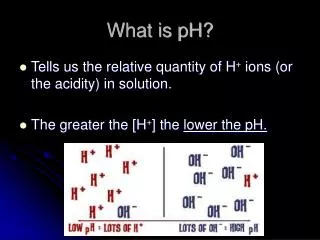
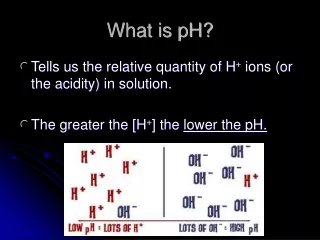
pH measures the amount of H+’s in a solution Water splits to form ions H+ and OH- What is pH? H+ H+ H+ H+ H+ H+
<7 on pH scale Higher concentration of H+ in solution than OH- Properties Sour taste Ability to turn litmus paper from blue to red Proton donor Acid More acidic Less acidic
Basic/Alkaline • >7 on pH scale • More OH-, Less H+ • Properties • Slippery • Bitter • Proton recipient Alkaline Less Alkaline
Neutral • pH of 7 • Water! • Balance of H+ and OH- ions
Each decrease in number (more acidic) represents a 10x increase in the number of H+ ions Interpreting the pH Scale
Testing pH • Indicator: Chemical that changes color • Test to determine pH • Litmus Test • Positive Acid Test: Turn blue litmus paper to red • Positive Alkaline Test: Turn red litmus paper to blue • pH Paper • Changes color • Match color to key to determine the pH