Understanding Mole Calculations: From Weight to Mole Ratios in Chemistry
150 likes | 308 Vues
This guide covers essential mole calculations critical for understanding chemical reactions and compositions. It explains how to convert weights, molecule counts, and moles using Avogadro’s number. Examples include calculating the molecules of O2 under specific conditions, the mass of detected selenium atoms on Mars, and determining the composition of glucose (C6H12O6). The content also discusses the relationship between moles and atomic/molecular weights, providing practical educational exercises for students in chemistry courses.

Understanding Mole Calculations: From Weight to Mole Ratios in Chemistry
E N D
Presentation Transcript
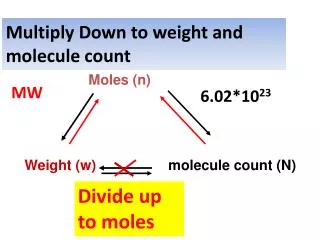
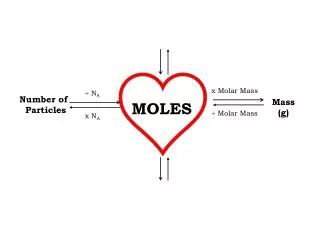
Multiply Down to weight and molecule count Moles (n) Weight (w) molecule count (N) MW 6.02*1023 Divide up to moles
Micro chip manufacture requires nearly oxygen-free conditions wherein the concentration of O2is at or below 17 pg/L. Given that the atomic mass of O is16g/mol, about how many molecules of O2 /L does this represent ? Note: 1 mol count=6.022*1023 • 5.4*1024 • 3.5*1022 • 5.3*1022 • 3.2*1011 • 6.4*1012 • 1.9*1013 • My answer isn’t above
A sample of 500 atoms of Se are detected by the Mars Rover while traversing the Sinus Miridani plain. What mass of Se was detected by the Rover ? (Se at. Wt.=79 g/mol. Assume 1 mol =6.02*1023 molecules) • 1.52*1020 g • 9.52*10-20 g • 1.051*10-23 g • 6.56*10-20 g • None of the above
You’ve stolen the Mars Rover’s atom detector and discovered 3.01*1020 atoms of a shiny metal in your backyard near the barbecue. The metal sample weighs 13.5 mg. What is the metal ? Note: 1 mol count=6.02*1023 molecules • Au=gold (197 g/mol) • Al (27 g/mol) • Cu (63.5 g/mol) • Fe (55.8 g/mol) • Pb (207 g/mol) • Problem is impossible to solve
Moles: part 2body parts (mole ratio) math: the knee bone is connected to the thigh bone….
How many hands??? 1 6 5 2 3 4 7 9 8 10 11 = 22 hands 2 hands 1 people x 11 people
How many toes connected to the hands (assuming no deformities) ? 5 4 3 2 1 x 10 toes person 5 hands 1 person hand = 50 toes x
Mole parts (ratios) math given 6 moles C3H4O6 How many moles of C ? 6 mol C3H4O6x 3moles C = 1 mol C3H4O6 18 moles C
Mole parts (ratio) math continued Given 45 grams of C6H12O6 …and MW of C6H12O6 =180 g/mol How many grams of C are present ?? First, convert mass to moles (all roads lead through moles) x 1 mole C6H12O6 180 g C6H12O6 45 g C6H12O6 = 0.25 moles C6H12O6
Mole parts (ratio) math continued Next, relate given moles to desired moles via mole ratio (need C moles ) 6 mole C 1 mole C6H12O6 0.25 moles C6H12O6 x ??? =1.5 moles C Finally, convert moles to grams in usual way: 12 g C 1 mole C x 1.5 moles C = 18 g C
Solving Mole parts (ratio) problems • Convert any given weight or molecule count to moles (“all roads lead through moles”) • Relate the computed moles above to desired moles • If necessary, convert desired moles to target grams or molecule count
In-class exercise #3b: chem1114 MoleCalculations: part 2 3.1)mole to mole: how many moles of O are present in 0.1666 mole of C6 H12O6? 1 mol O 3.2) mole to mole how many moles of C6H12O6 can be made with 12 mole of O ? 2moles C6H12O6
In-class exercise #7b: chem 1114 Mole Calculations: part 2 3.3) weight to moles how many moles of C6H12O6 in a sample containing 216 g C ? 3mol 3.4)moles to weight how many grams of C6H12O6 are formed with 0.2666 mol H? = 4 g
3.5) moles to molecules how many molecules of O are present in 0.8333 mol of C6 H12O6? =5*1023 3.6) molecules to moles how many moles of C6 H12O6 are formed from 5.04*1025 atoms of H ? 7 moles
3.7 mass to molecules: how many moleculesof C6 H12O6form from 96 g of C ? 8 *1023 molecules ofC6H12O6 3.8 atoms to mass: how many grams of H are combined with 2.7*1024 atoms of O in C6H12O 6 ? 9 grams H