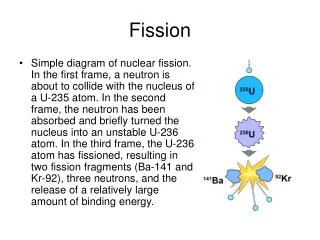
Fission
Fission. Readings: Modern Nuclear Chemistry, Chapter 11; Nuclear and Radiochemistry, Chapter 3 General Overview of Fission Energetics The Probability of Fission Fission Product Distributions Total Kinetic Energy Release Fission Product Mass Distributions


Fission
E N D
Presentation Transcript
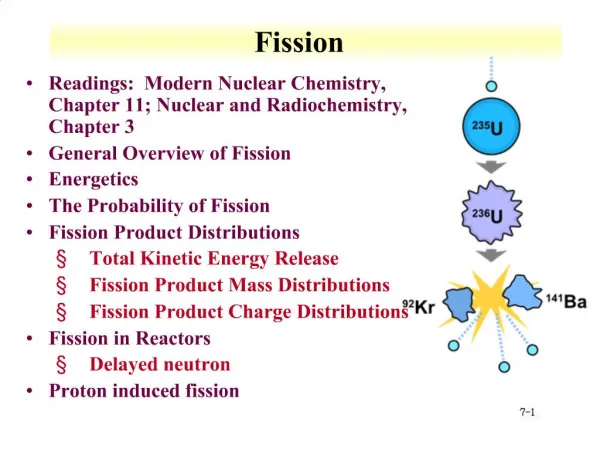
Fission • Readings: Modern Nuclear Chemistry, Chapter 11; Nuclear and Radiochemistry, Chapter 3 • General Overview of Fission • Energetics • The Probability of Fission • Fission Product Distributions • Total Kinetic Energy Release • Fission Product Mass Distributions • Fission Product Charge Distributions • Fission in Reactors • Delayed neutron • Proton induced fission
Nuclear Fission • Fission discovered by Otto Hahn and Fritz Strassman, Lisa Meitner in 1938 • Demonstrated neutron irradiation of uranium resulted in products like Ba and La • Chemical separation of fission products • For induced fission, odd N • Addition of neutron to form even N • Pairing energy • In 1940 G. N. Flerov reported that 238U undergoes fission spontaneously • half life of round 1016 y • Several other spontaneous fission isotopes found • Z > 90 • Partial fission half lives from nanoseconds to 2E17 years
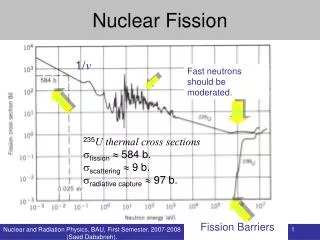
Fission • Can occur when enough energy is supplied by the bombarding particle for the Coulomb barrier to be surmounted • Fast neutron • Proton • Spontaneous fission occurs by tunneling through barrier • Similar to alpha decay • Thermal neutron induced fission from pairing of unpaired neutron • Nuclides with odd number of neutrons fissioned by thermal neutrons with large cross sections • follow 1/v law at low energies, sharp resonances at high energies
Energetics Calculations • Why does 235U undergo neutron induced fission for thermal energies? • Where does energy come from? • Generalized energy equation • AZ + n A+1Z + E • For 235U • E=(40.914+8.071)-42.441 • E=6.544 MeV • For 238U • E=(47.304+8.071)-50.569 • E=4.806 MeV • For 233U • E=(36.913+8.071)-38.141 • E=6.843 MeV • Fission requires around 5-6 MeV
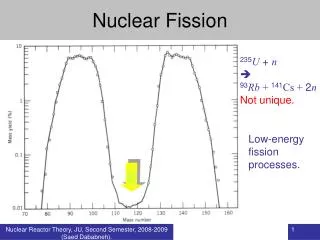
Usually asymmetric mass split MH/ML1.4 for uranium and plutonium due to shell effects, magic numbers Heavy fragment peak near A=132, Z=50, N=82 Symmetric fission is suppressed by at least two orders of magnitude relative to asymmetric fission Occurs in nuclear reactions Competes with evaporation of nucleons in region of high atomic numbers As mass of the fissioning system increases Location of heavy peak in the fission remains constant position of the light peak increases Fission Process Nucleus absorbs energy Excites and deforms Configuration “transition state” or “saddle point” Nuclear Coulomb energy decreases during deformation Nuclear surface energy increases Saddle point key condition rate of change of the Coulomb energy is equal to the rate of change of the nuclear surface energy Induces instability that drives break up of nucleus Fission Process
Fission Process • If the nucleus deforms beyond this point it is committed to fission • Neck between fragments disappears • Nucleus divides into two fragments at the “scission point.” • two highly charged, deformed fragments in contact • Large Coulomb repulsion accelerates fragments to 90% final kinetic energy within 10-20 s
Fission Products • Fission yield curve varies with fissile isotope • 2 peak areas for U and Pu thermal neutron induced fission • Variation in light fragment peak • Influence of neutron energy observed 235U fission yield
Fission • Primary fission products always on neutron-excess side of stability • high-Z elements that undergo fission have much larger neutron-proton ratios than the stable nuclides in fission product region • primary product decays by series of successive - processes to its stable isobar • Yields can be determined • Independent yield: specific for a nuclide • Cumulativeyield: yield of an isobar • Beta decay to valley of stability • Data for independent and cumulative yields can be found or calculated • For reactors • Emission of several neutrons per fission crucial for maintaining chain reaction • “Delayed neutron” emissions important in control of nuclear reactors Comparison of cumulative and independent yields for A=141 http://www-nds.iaea.org/sgnucdat/c2.htm
Fission products • Particles form more spherical shapes • Converting potential energy to emission of “prompt” neutrons • Gamma emission after neutrons • Then decay • Occasionally one of these decays populates a high lying excited state of a daughter that is unstable with respect to neutron emission • “delayed” neutrons • 0.75 % of total neutrons • 137-139I and 87-90Br as examples
Delayed Neutrons • Fission fragments are neutron rich • More neutron rich, more energetic decay • In some cases available energy high enough for leaving the residual nucleus in such a highly excited state • Around 5 MeV • neutron emission occurs
Delayed Neutrons in Reactors • Control of fission • 0.1 msec for neutron from fission to react • Need to have tight control • 0.1 % increase per generation • 1.001^100, 10 % increase in 10 msec • Delayed neutrons useful in control • Longer than 0.1 msec • 0.65 % of neutrons delayed from 235U • 0.26 % for 233U and 0.21 % for 239Pu • Fission product poisons influence reactors • 135Xe capture cross section 3E6 barns
Nuclear reactors • Probable neutron energy from fission is 0.7 MeV • Fast reactors • High Z reflector • Thermal reactors need to slow neutrons • Water, D2O, graphite • Low Z and low cross section • Power proportional to number of available neutrons • Should be kept constant under changing conditions • Control elements and burnable poisons • k=1 (multiplication factor) • Ratio of fissions from one generation to the next • k>1 at startup
Fission Process and Damage • Neutron spatial distribution is along the direction of motion of the fragments • Energy release in fission is primarily in the form of the kinetic energies • Energy is “mass-energy” released in fission due to the increased stability of the fission fragments • Recoil length about 10 microns, diameter of 6 nm • About size of UO2 crystal • 95 % of energy into stopping power • Remainder into lattice defects • Radiation induced creep • High local temperature from fission • 3300 K in 10 nm diameter
Energetics • Any nucleus of A> 100 into two nuclei of approximately equal size is exoergic. • Why fission at A>230 • Separation of a heavy nucleus into two positively charged fragments is hindered byCoulomb barrier • Treat fission as barrier penetration • Barrier height is difference between • Coulomb energy between the two fragments when they are just touching • the energy released in the fission process • Near uranium both these quantities have values close to 200 MeV
Energetics • 200Hg give 165 MeV for Coulomb energy between the fragments and 139 MeV for energy release • Lower fission barriers for U when compared to Hg • Barrier height increases more slowly with increasing nuclear size compared to fission decay energy • Spontaneous fission is observed only among the very heaviest elements • Half lives generally decrease rapidly with increasing Z
Energetics • Generalized Coulomb barrier equation • Compare with Q value for fission • Determination of total kinetic energy • Equation deviates at heavy actinides (Md, Fm) • Consider fission of 238U • Assume symmetric • 238U119Pd + 119Pd + Q • Z=46, A=119 • Vc=462*1.440/(1.8(1191/3)2)=175 MeV • Q=47.3087-(2*-71.6203) = 190.54 MeV • asymmetric fission • 238U91Br + 147La + Q • Z=35, A=91 • Z=57, A=147 • Vc=(35)(57)*1.44/(1.8*(911/3+1471/3))=164 MeV • Q=47.3087-(-61.5083+-66.8484) = 175.66 MeV • Realistic case needs to consider shell effects
Fission Isomers • Some isomeric states in heavy nuclei decay by spontaneous fission with very short half lives • Nano- to microseconds • Fissioning isomers are states in these second potential wells • Also called shape isomers • Exists because nuclear shape different from that of the ground state, • Around 30 fission isomers are known, from U to Bk • Induced by neutrons, protons, deuterons, and aparticles
Fission Isomers: Double-humped fission barrier • At lower mass numbers, the second barrier is rate-determining, whereas at larger A, inner barrier is rate determining • Symmetric shapes are the most stable at two potential minima and the first saddle, but some asymmetry lowers second saddle
Proton induced fission • Energetics impact fragment distribution • excitation energy of the fissioning system increases • Influence of ground state shell structure of fragments would decrease • Fission mass distributions shows increase in symmetric fission
Topic Review • Mechanisms of fission • What is occurring in the nucleus during fission • Understand the types of fission • Particle induced • Spontaneous • Energetics of fission • Q value and coulomb barrier • The Probability of Fission • Cumulative and specific yields • Fission Product Distributions • Total Kinetic Energy Release • Fission Product Mass Distributions
Questions • Compare energy values for the symmetric and asymmetric fission of 242Am. • What is the difference between prompt and delayed neutrons in fission. • What is the difference between induced and spontaneous fission. • What influences fission product distribution? • Compare the Coulomb barrier and Q values for the fission of Pb, Th, Pu, and Cm. • Describe what occurs in the nucleus during fission. • Compare the energy from the addition of a neutron to 242Am and 241Am. Which isotope is likely to fission from an additional neutron.
Pop Quiz • Provide calculations showing why 239Pu can be fissioned by thermal neutron but not 240Pu. • Compare the Q value and Coulomb energy (Vc) from the fission of 239Pu resulting in 138Ba and 101Sr. Is this energetically favored?