Alkene Reaction Mechanisms for Organic Chemists
170 likes | 205 Vues
Explore key alkene reaction mechanisms, including hydrogenation, halogenation, hydration, and polymerization. Learn about the intricate details of addition reactions and unsymmetrical additions. Enhance your knowledge in organic chemistry.

Alkene Reaction Mechanisms for Organic Chemists
E N D
Presentation Transcript
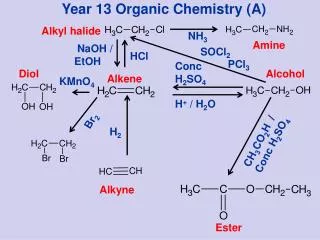
Alkene Reactions • Addition Reactions - only one product • 1. Hydrogenation (H2): Alkene + Hydrogen --> Alkane • 2. Halogenation (X2):Alkene + diatomic halogen --> Dihaloalkane • 3. Halgenation (HX):Alkene + HX --> Haloalkane • 4. Hydration (HOH):Alkene + HOH --> alcohol • 5. Polymerization:Many alkenes add together into a long alkane chain
1. Hydrogenation Reaction • Alkene + Hydrogen --> Alkane • Hydrogen molecule adds to carbons with double bond.
2. Halogenation Reaction • Alkene + Halogen --> Dihaloalkane • Halogen molecule adds to carbons with double bond.
Unsymmetrical Addition Reactions • Markovnikov’s Rule • For double bonds that have unequal numbers of hydrogen atoms attached. • For unsymmetrical reactants such as HX and HOH • The hydrogen of the reactant goes to the carbon of the double bond that already has the most hydrogen atoms. “the rich get richer” • The -X or the -OH go the other carbon (the one with the least amount of hydrogen
3. Halogenation Reaction • Alkene + HX --> haloalkane • HCl molecule adds to carbons with double bond.
4. Hydration Reaction • Alkene + HOH --> alcohol • HOH molecule adds to carbons with double bond.
5. Polymerization Reaction • Alkene + Alkene + Alkene --> long chain of carbons • Double bonds convert to single bonds
What is the structure for Alkene Reactions