Electrochemistry
This chapter covers the essential concepts of oxidation numbers and redox reactions, as delineated in the IB curriculum. It begins with defining oxidation as the loss of electrons and reduction as the gain of electrons, encapsulated in the mnemonic "LEO says GER". Through detailed examples, it explains how to assign oxidation numbers and determines which elements are oxidized and reduced in various reactions. Additionally, the chapter provides systematic methods for balancing redox equations, ensuring accurate stoichiometry in complex chemical reactions.

Electrochemistry
E N D
Presentation Transcript
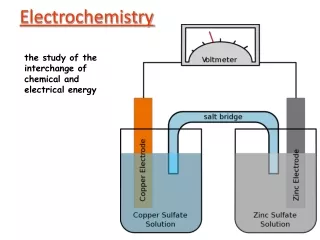
Electrochemistry Textbook Chapter 20 IB Topics 9 and 19
Oxidation Numbers • IB Topic 9.1.1- 9.1.4 • Text p. 843-846 • Oxford p. 54. 55
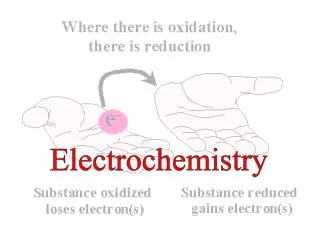
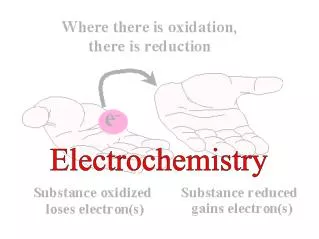
LEO says GER!
GER! LEO says Loss of Electrons = Oxidation Gain of Electrons =Reduction
Reduction Is Gain (of electrons) • Oxidation Is Loss (of electrons)
Oxidation Numbers • Oxidation/Reduction (ReDox)-- Whenever a substance loses electrons and another substance gains electrons • Oxidation Numbers--system used to keep track of electron transfers
Using Oxidation Numbers • Careful examination of the oxidation numbers of atoms in an equation allows us to determine what is oxidized and what is reduced in an oxidation-reduction (Redox) reaction
An increase in the oxidation number indicates that an atom has lost electrons and therefore oxidized. A decreasein the oxidation number indicates that an atom has gained electrons and therefore reduced Example Zn + CuSO4 ZnSO4 + Cu 0 +2 +6-2 +2+6-2 0 Zn: 0+ 2Oxidized Cu: +20Reduced Using Oxidation Numbers
Exercise For each of the following reactions find the element oxidized and the element reduced Cl2 + KBr KCl + Br2 Cu + HNO3 Cu(NO3)2+ NO2 + H2O HNO3 + I2 HIO3 + NO2
Exercise For each of the following reactions find the element oxidized and the element reduced Cl2 + KBr KCl + Br2 0 +1-1 +1-10 Br increases from –1 to 0 -- oxidized Cl decreases from 0 to –1 -- Reduced K remains unchanged at +1
Exercise For each of the following reactions find the element oxidized and the element reduced Cu + HNO3 Cu(NO3)2+ NO2 + H2O 0 +1+5-2 +2 +5-2 +4 –2 +1-2 • Cu increases from 0 to +2. It is oxidized • Only part of the N in nitric acid changes from +5 to +4. It is reduced • The nitrogen that ends up in copper nitrate remains unchanged
Exercise For each of the following reactions find the element oxidized and the element reduced HNO3 + I2 HIO3 + NO2 1 +5 -2 0 +1+5-2+4-2 • N is reduced from +5 to +4. It is reduced • I is increased from 0 to +5 It is oxidized • The hydrogen and oxygen remain unchanged.
Assignment • Oxidation Numbers/Naming Review packet
Balancing Redox Reactions • IB Topics 9.2.1-9.2.4 • Text p. 846-850 • Oxford p. 55
Balancing Redox Reactions • Many Redox rxns are complex and difficult to balance . • A systematic approach to balancing these reaction is required.
MnO41- (aq) + C2O42- (aq) Mn2+(aq) + CO2(aq) Balancing Redox Equations 1 • Divide the equation into 2 half reactions—one for oxidation, one for reduction. • Balance each half reaction • Balance elements other than H and O • Balance O by adding H2O as needed • Balance H by adding H+ as needed.(acidic solution) • Balance charge by adding e- as needed. • Multiply half reactions by integers so that the # of e- lost in one reaction = # of e- gained in the other reaction. • Add the two half reactions. Simplify by canceling species that appear on both sides of the arrow. • Check your work. Make sure that both the atoms and charges balance
Cr2O72- (aq) + Cl1- (aq) Cr 3+ (aq) + Cl2 (g) • Divide the equation into 2 half reactions—one for oxidation, one for reduction. • Balance each half reaction • Balance elements other than H and O • Balance O by adding H2O as needed • Balance H by adding H+ as needed. .(acidic solution) • Balance charge by adding e- as needed. • Multiply half reactions by integers so that the # of e- lost in one reaction = # of e- gained in the other reaction. • Add the two half reactions. Simplify by canceling species that appear on both sides of the arrow. • Check your work. Make sure that both the atoms and charges balance Balancing Redox Equations 2
Divide the equation into 2 half reactions—one for oxidation, one for reduction. • Balance each half reaction • Balance elements other than H and O • Balance O by adding H2O as needed • Balance H by adding H+ as needed. .(acidic solution) • Balance charge by adding e- as needed. • Multiply half reactions by integers so that the # of e- lost in one reaction = # of e- gained in the other reaction. • Add the two half reactions. Simplify by canceling species that appear on both sides of the arrow. • Check your work. Make sure that both the atoms and charges balance Balancing Redox Equations 3 Cu (s) + NO31- (aq) Cu 2+(aq) + NO2(aq)
Divide the equation into 2 half reactions—one for oxidation, one for reduction. • Balance each half reaction • Balance elements other than H and O • Balance O by adding H2O as needed • Balance H by adding H+ as needed. .(acidic solution) • Balance charge by adding e- as needed. • Multiply half reactions by integers so that the # of e- lost in one reaction = # of e- gained in the other reaction. • Add the two half reactions. Simplify by canceling species that appear on both sides of the arrow. • Check your work. Make sure that both the atoms and charges balance Balancing Redox Equations 4 Mn2+ (aq) + NaBiO3 (s) Bi 3+(aq) + MnO4 1- (aq) + Na1+(aq)
Text p. 884 • Text p. 884 • Problems # 20a, b, c, d 22 a, b, c, d “acidic solution only”