Subatomic Particles
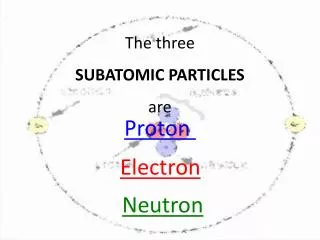
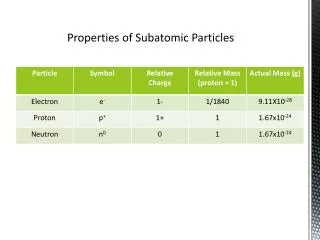
Subatomic Particles. A.K.A. particles that are smaller than an atom. p +. +1. nucleus. Attract electrons. ~1amu**. Buffer p+ to p+ repulsions to help stabilize the nucleus. 0. n. ~1amu. nucleus. Electron clouds around the nucleus. 1/2000 amu. Participate in chemical reactions. e-.


Subatomic Particles
E N D
Presentation Transcript
Subatomic Particles A.K.A. particles that are smaller than an atom. p+ +1 nucleus Attract electrons ~1amu** Buffer p+ to p+ repulsions to help stabilize the nucleus 0 n ~1amu nucleus Electron clouds around the nucleus 1/2000 amu Participate in chemical reactions e- -1 * protons and neutrons are collectively called nucleons because they are located in the nucleus. ** Amu (atomic mass unit) is defined as 1/12 of the atomic mass of the carbon-12 isotope (1.6605402 x 10-27 kg)
If a football stadium represented the size of an atom (the electron clouds surrounding the nucleus), the nucleus is estimated to be about the size of a nickel.
Atomic Number and Mass Number Atomic number – number of protons in an atom. Also indicates the name of the element (Periodic Table). Mass number – number of nucleons (protons and neutrons) in an atom. It is always a whole #, never a decimal approximation. It is specific to a particular atom, therefore it is not on the Periodic Table.
Isotopes Isotopes – atoms that have the same number of protons but different number of neutrons Hyphen NotationandSymbol Notationfor atoms. Mass number C 14 Carbon-14 6 Atomic number Therefore it has 8 neutrons
Atomic Mass Atomic Mass – average of all naturally occurring isotopes. Be careful not to confuse atomic mass and mass number. It is the number found on the Periodic Table. Atomic Number Based on this atomic mass, the most abundant Carbon isotope would probably be Carbon-12. Atomic mass
Ions Ions are atoms that have become charged due to an imbalance between protons and electrons. Ions can only be formed by the loss or gain of electrons. The loss or gain of protons, on the other hand does not form ions because if protons are lost or gained, the nucleus changes (nuclear reaction) and the atom becomes a new element. Cation Anion Is an atom that has LOST electrons and become POSITIVELY charged. Is an atom that has GAINED electrons and become NEGATIVELY charged. The “t” in the word cation looks like a plus sign, therefore it should help you remember that it is positively charged.
Symbols for Ions Symbols for ions are simply the elements symbol with a superscript to indicate the charge of the ion. Chlorine -1 anion Sodium +1 cation - + Na Cl Lead +4 cation +4 Pb
Summary of Terms • Ion-an atom that has become charged due to a loss or gain of electrons. (cations and anions) • Atomic Number-indicates the # protons as well as the # electrons if the atom is neutral. It also indicates the identity of the element. • Mass Number-thesum of protons plus neutrons. This number is specific to an individual atom. • Atomic Mass-the average of mass #’s for all the naturally occurring isotopes for a given element. • Isotopes-atoms of the same element (same # protons) that have different # of neutrons.
Applying the Concepts Indicate the number of protons, neutrons and electrons for each species below. Also indicate whether each species is cation, anion, or an atom. # protons # neutrons # electrons species 47 63 47 atom atom 77 123 77 82 139 78 +4 cation Silicon-26 14 12 14 atom
More Examples # protons # neutrons # electrons species 1 0 1 atom 24 +2 cation 26 35 atom 40 49 40 Gold-198 79 119 79 atom 74 105 68 +6 cation
The Periodic Table the organization of elements into groups by similar chemical properties
Periodic Classification Groups/families – the vertical columns of the periodic table Periods – the horizontal rows of elements in the periodic table Group/Family Period
Types of Elements Metals– an element that is a good electrical conductor and a good heat conductor. Commonly form cations. Nonmetals– an element that is a poor conductor of heat and electricity. Commonly form anions. Metalloids– an element that has some characteristics of metals and some characteristics of nonmetals Copper: Metal Sulfur: Nonmetal Silicon: Metalloid
Groups All groups have similar chemical properties Group 1: Alkali Metals – have a silvery appearance and are very soft in pure state. Very reactive. Not found in nature as a free element. Generally form +1 cations in ionic compounds. They like to bond with anions. Group 2: Alkaline-Earth – harder, denser, and stronger than alkali metals. Fairly reactive. Not found in nature as a free element. Generally form +2 cations in ionic compounds. Groups 3-12: Transition Metals – good conductors of electricity, high luster. Less reactive than alkali/alkaline-earth. Cation charges vary. Rubidium: Alkali Magnesium: Alkaline-Earth Gold: Transition
Groups (continued) Group 16: Chalcogens– include non-metals and metalloids. Generally form –2 anions in ionic compounds. Group 17: Halogens– most reactive nonmetals. Combine with most metals to form salts. Generally form –1 anions in ionic compounds. Group 18: Noble Gases– inert gases. I2 2 2
1 18 13 14 15 16 17 2 3 4 5 6 7 8 9 10 11 12 Notice the labeling scheme for the groups on this periodic table. Roman numerals and capital letters are used. Groups with capital letter A are considered the representative elements. The more recent labeling scheme is to simply number them from 1 to 18 from left to right
Chemical Properties Elements in the same Group/Family will have similar chemical properties. For example if Lithium combines with chlorine to form LiCl, then all the other alkali metals will react similarly. NaCl, KCl, RbCl, CsCl Bromine readily reacts with magnesium to form MgBr2, then all the other halogens will react similarly. MgF2, MgCl2, MgI2
The Mole A mole, in chemistry, is an SI base unit for counting how many. It is similar to a dozen because a dozen indicates “how many”. In fact a dozen things = 12 of those things. Except in chemistry atoms and molecules are very small, therefore we would need a lot of them to observe properties about a substance. A dozen atoms or a dozen molecules is just not practical, therefore we need a much larger #.
The Mole, cont. Avogadro’s number A mole means 6.02 x1023 “things” The things are generally atoms, molecules or ions. It is similar to a dozen in that it indicates “how many”, 1 dozen atoms = 12 atoms Similarly,1 mole atoms = 6.02 x1023 atoms It is similar to a dozen, 1 dozen molecules = 12 molecules Similarly,1 mole molecules = 6.02 x1023 molecules
The Mole, cont. *IMPORTANT* Would a dozen pencils have the same mass as a dozen people? OF COURSE NOT! Then the same concept applies to moles. 1 mole CO2, carbon dioxide, does not have the same mass as 1 mole Pb(NO3)4, Lead(IV) Nitrate WHY? Simply because 1 CO2 molecule has less mass than 1 molecule of Pb(NO3)4. Therefore 6.02 x1023 CO2 molecules (1mole) will obviously have less mass than 6.02 x1023 Pb(NO3)4 molecules (1mole) .
Molecular Weight and Molar Mass Molecular Weight (A.K.A. Formula Weight for ionic compounds) and Molar Mass are calculated the same way, the only difference is the unit and meaning. Molecular Weight is the weight of a molecule measured in amu. Molar Mass is the mass (in grams) of one mole of a substance.
Calculating Molecular Weight and Molar Mass Example: Calculate the molecular weight of Pb(NO3)2. = X Pb 207.20 1 207.20 N 14.01 2 28.02 O 16.00 6 96.00 331.22 amu Therefore the molecular weight is 331.22 amu, therefore the molar mass or 331.22 g/mol. (Same # different unit and meaning)
Mole Conversions Converting between moles, grams and # of particles (atoms, molecules or ions.) Use Dimensional Analysis Given Quantity and Unit Conversion Factor(s) Desired Quantity And Unit = X
The Concept Map for Mole Conversions # of Particles (Atoms, molecules) Avogadros # Ratio of atoms to 1 molecule 1 mole = 6.02 E23 “things” moles Depends on whetherthe smallest unit of the substance is a molecule or an atom. # atoms Molar Mass # grams = 1 mole Mass (grams)
Examples 1) How many moles of H3PO4 are in 9.204 g H3PO4? 9.204 g H3PO4 X 0.09392 mol H3PO4 2) Calculate the mass of 0.109 mol of Fe(OH)3. 0.109 mol Fe(OH)3 X 11.6 g Fe(OH)3
More Examples 3) How many particles are in 50.90 mol H2O? What kind of particles are they (desired unit)? MOLECULES 50.90 mol H2O 3.064 E25 H2O molecules X 4) How many oxygen atoms are there in 5.60 mol of Pb(SO4)2? 5.60 mol Pb(SO4)2 X X 2.70 E25 Oxygen atoms
More Examples 5) How many molecules of H2O are in 603.8 g H2O? 603.8 g H2O X X = 2.017 E25 molec H2O 6) What is the mass of 9.08E24 Br2 molecules ? 9.08E24 Br2 molec X X = 2.41 E3 g Br2
Valence Electrons • Valence electrons are the electrons in the outermost energy shell, or valence shell. • For representative elements (groups 1-2 and 13-18) the group # will help you determine how many valence electrons an atom has.
The Octet Rule • The representative elements, when forming ions, will attempt to have 8 electrons in their outer shell. • Atoms will either lose or gain electrons to result in a completed outer shell.(8 electrons)
Valence Electrons The Group # (for the representative elements only) indicates the number of valence electrons. For example all elements in Group 1 have 1 valence electron. Therefore, they can either gain 7 electron or lose 1 electron to result with a completed valence shell. Obviously it will require less energy to lose 1 than to try to gain 7, therefore all Group 1 metals become a +1 cation as a result of losing 1 electron.
Common Ion Formation (most representative elements) • Group 17 needs to gain 1 electron becoming a -1anion. • Group 16 needs to gain 2 electrons becoming a -2anion. • Group 15 (non-metals only) needs to gain 3 electrons becoming a -3anion. • Group 14 (non-metals only) can either gain 4 or lose 4 electrons therefore can form +4 cations or -4 anions. • Group 13 (non-metals only) needs to lose 3 electrons becoming a +3cation. • Group 1 needs to lose 1 electron becoming a +1cation. • Group 2 needs to lose 2 electrons becoming a +2cation.
Common Ion Formation (transition metals) • All metals form cations. • Transitions metals can form multiple charges depending upon the situation. • The most common charges (listed with the most common first) are the little numbers to the right of the element symbol on your Periodic Table.
Examples Write the symbol for the most common charge if the following atoms were to become ions. Na S Cr Sn Br Sr Al Na+1 S-2 Cr+6 Sn+4 Br-1 Sr+2 Al+3