Periodic Table in Applied Chemistry
Explore the basics of substances, the development of the Periodic Table, properties of metals and nonmetals, and the division between metal and nonmetal elements in this informative guide.

Periodic Table in Applied Chemistry
E N D
Presentation Transcript
Ch. 14 Periodic Table Applied Chemistry Milbank High School
Substances • __________- simplest kind of matter • all one kind of atom. • __________ are substances that can be broken down only by chemical methods • When broken down, the pieces have completely different properties than the original compound. • Made of two or more atoms
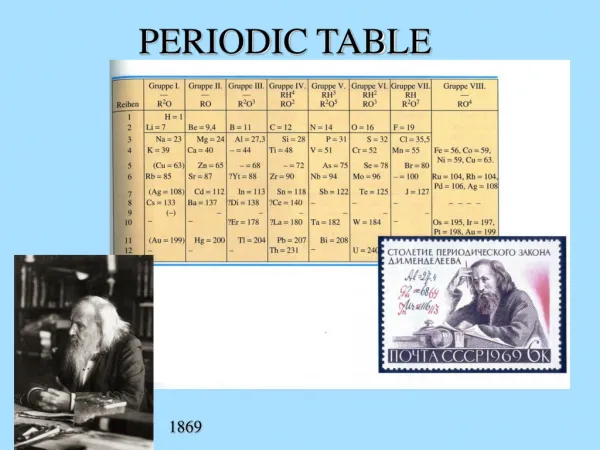
Development of the Periodic Table • mid-1800s, about ___ elements • Dmitri Mendeleev – Russian chemist • Arranged elements in order of increasing ___________ • Thus, the first “Periodic Table” • Left blanks for undiscovered ________ • When discovered, good prediction
Periodic table • Horizontal rows = _______ • There are __ _______ • Periodic law: • Vertical column = _______ (or family) • Similar physical & chemical prop. • Identified by number & letter
Metals • _________ – alkali metals • Group 2A – _______________ • Transition metals and _____ _________ metals • All metals are solids at room temperature, except _____.
Nonmetals • ___________: generally nonlustrous, poor conductors of electricity • Some gases (O, N, Cl); some are brittle solids (S); one is a fuming dark red liquid (Br) • Group 7A – _________ • Group 8 – ______ _______
Division between metal & nonmetal • Heavy, stair-step line • ____________ border the line • Properties intermediate between metals and nonmetals • Learn the general behavior and trends of the elements, instead of memorizing each element property