Hydrogen-Bond Catalysis
Hydrogen-Bond Catalysis. For reviews see: M. S. Taylor, E. N. Jacobsen, Angew. Chem. Int. Ed. 2006, 45 , 1520-1543 A. G. Doyle, E. N. Jacobsen, Chem. Rev., 2007 , 5713-5743. Nadia Fleary-Roberts 03/02/10. A brief history…. Lewis acids has dominated enantioselective catalysis

Hydrogen-Bond Catalysis
E N D
Presentation Transcript
Hydrogen-Bond Catalysis For reviews see: M. S. Taylor, E. N. Jacobsen, Angew. Chem. Int. Ed. 2006, 45, 1520-1543 A. G. Doyle, E. N. Jacobsen, Chem. Rev., 2007, 5713-5743 Nadia Fleary-Roberts 03/02/10
A brief history… • Lewis acids has dominated enantioselective catalysis • acceleration of Diels Alder reaction with AlCl3 Yates P.; Eaton P., J.Am. Chem. Soc. 1960, 82, 4436 • Wynberg reported asymmetric conjugate addition reactions with cinchona alkaloids • bearing a free hydroxyl group. Hiemstra H.; Wynberg H., J. Am. Chem. Soc. 1981, 103, 417-430
Jacobsen reported asymmetric hydrocyanation of aliphatic and aromatic aldehydes Sigman, M. S.; Jacobsen, E. N. J. Am. Chem. Soc. 1998, 120, 4901
Lewis acid catalysis • Highly tunable • Can vary counter ion, chiral ligand • Lewis base/acid interactions are stronger • Interactions are more directional H-bond catalysis Moderately tunable Active catalyst Potentially recoverable
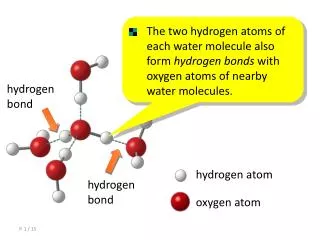
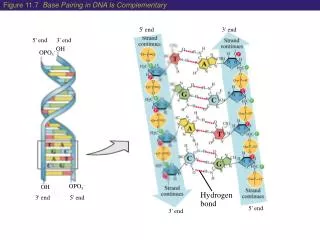
Strength can vary from 0.4 to 40 kcalmol-1. What is a hydrogen bond? “An XH···A interaction is called a ,hydrogen bond', if 1. it constitutes a local bond, and 2. XH acts as a proton donor to A.” Steiner, Angew. Chem. Int. Ed. 2002, 41, 48 Role of H-bonds: DNA base pairing Ligand/receptor binding G.A. Jeffrey, An Introduction to Hydrogen Bonding, Oxford University Press, New York, 1997 Most Hydrogen bonding in H-bond catalysis is of moderate strength
Brønsted acids can accelerate organic reactions by either of two fundamental mechanisms: • protonation of the electrophile in a prior to nucleophilic attack • proton transfer to the transition state in the rate-determining step Specific acid catalysis General acid catalysis
Modes of bonding Double H-bond donors Increased strength and directionality e.g. Ureas, thioureas, Guanidinium and Amidinium ions X = O, S
Single H-bond donors • Less strength than double H-bond donors • Less directionality • e.g. Diols, biphenols, chiral phosphoric acids
Bifunctional catalysis • a single or dual H-bond donor site flanked by sites for secondary interaction with substrates. e.g. Proline and proline analogs, cinchona alkaloids and derivatives
Double H-bond donors Ureas and Thioureas b R1 = Bn, R2 = H, R3 = OCH3, X= S cR1 = Bn, R2 = H, R3 = OCOtBu, X = O d R1 = R2 = CH3, R3 = OCOtBu, X =S eR1 = Bn, R2 = CH3, R3 = tBu, X = S • Originally developed as ligands for lewis acidic metals. • Most applicable class of chiral H-bond donors • Can promote addition of a range of nucleophiles • Imine activation
Double H-bond donors • Diels Alder reaction Wittkopp A.; Schreiner P. R., Chem. Eur. J. 2003, 9, • Aza Baylis-Hillman reaction eR1 = Bn, R2 = CH3, R3 = tBu, X = S Raheem, I. T.; Jacobsen, E. N. AdV. Synth. Catal. 2005, 347, 1701
Double H-bond donors • Asymmetric Strecker reaction • Scaleable catalytic synthesis • Imines derived from alky, aryl, heteroaryl aldehydes • Aqueous cyanide salts • Robust catalyst • Access to the (R)-enantiomer of tert-leucine Zuend S.J.; Coughlin M. P.; Lalonde M. P.; Jacobsen E. N., Nature, 2009, 461, 968
Double H-bond donors • Asymmetric Pictet Spengler reaction Taylor, M. S.; Jacobsen, E. N. J. Am. Chem. Soc. 2004, 126, 10558
Bifunctional Thiourea derivatives • 1,4- additions Takemoto catalyst T. Okino, Y. Hoashi, Y. Takemoto, J. Am. Chem. Soc. 2003, 125, 12672 – 12673 B.-J. Li, L. Jiang, M. Liu, Y.-C. Chen, L.-S. Ding, Y. Wu, Synlett2005, 4, 603 • Aza-henry reaction-(addition of nitroalkanes to imines ) T. Okino, S. Nakamura, T. Furukawa, Y. Takemoto, Org. Lett. 2004, 6, 625
1,2-additions D. E. Fuerst, E. N. Jacobsen, J. Am. Chem. Soc. 2005, 127, 8964 • Other thiourea catalysts efficient catalyst for Baylis-Hillman reactions
Double H-bond donors Guanidinium and Amidinium ions • Capable of double H-bond interactions • Are positively charged so result in increased H-bond donor abiltity. • Strecker reaction T. Steiner, Angew. Chem. 2002, 114, 50; Angew. Chem. Int. Ed. 2002, 41, 48 .
Double H-bond donors Amidinium catalysed reactions • Nitro-Mannich reaction • Diels Alder reaction k Ar = 3,5-(CF3)2C6H3 Schuster T.; Bauch M.; Durner G.; Gmbel M. W.; Org. Lett. 2000, 2, 179 – 181.
Single H-bond donors Diols and Biphenols • Hetero Diels Alder reaction catalysed by chiral diols Huang Y.; Unni, A. K.; Thadani, A. N.; Rawal V.H., Nature, 2003, 424, 146 • Baylis-Hillman reaction -Binol derivatives
Single H-bond donors • Chiral phosphoric acids • Mannich reaction T. Akiyama, J. Itoh, K. Yokota, K. Fuchibe, Angew. Chem. 2004, 116, 1592 – 1594 Angew. Chem. Int. Ed. 2004, 43, 1566 –1568 D. Uraguchi, M. Terada, J. Am. Chem. Soc. 2004, 126, 5356 – 5357
Reductive amination HEH = ethyl Hantzsch ester R. I. Storer, D. E. Carrera, Y. Ni, D. W. C. Macmillan, J. Am. Chem. Soc. 2006, 128, 84
Bifunctional catalysis Proline catalysed reactions • enatioselective aldol cyclizations Z. G. Hajos, D. R. Parrish, (Hoffman-La-Roche), German Patent DE 2102623, 1971 [Chem. Abstr. 1972, 76, 59072];
Proposed mechanism for proline catalysed transformations A. G. Doyle, E. N. Jacobsen, Chem. Rev., 2007, 5713-5743
Proline analogues u higher yields and selectivity compared to proline x increased solubility Tetrazole is a pharmacore for carboxylic acid
Cinchona alkaloids Bifunctional catalysis • Enatioselective conjugate additions • Baylis-Hillman reaction Nakano A. et . al. 2006, 62, 381 Iwabuchi Y. et. al. J. Am. Chem. Soc. 1999, 121, 10219 Connon S.J., Chem. Comm., 2008, 2499
Bifunctional catalysis Oligiopeptides • Cyclo(l-phenylalanine-l-histidine) hydrocyanation of aldehydes Uncertainty concerning mechanism S. Inoue, J.-I. Oku, J. Chem. Soc. Chem. Commun. 1981, 229 –230
Summary A dynamic and large area of research which is continuing to be explored Applicable to a wide number of transformations
CAKE-OFF 2010!! Don’t forget to vote!!