Chromatography
Chromatography. Pn. Suryati Bt. Syafri 2008. LEARNING OUTCOME. After studying this topic student should be able to : Define chromatography Explain classification of chromatography technique Explain type of phase in chromatography

Chromatography
E N D
Presentation Transcript
Chromatography Pn. Suryati Bt. Syafri 2008
LEARNING OUTCOME After studying this topic student should be able to : • Define chromatography • Explain classification of chromatography technique • Explain type of phase in chromatography • Describe about paper chromatography (definition, principle, method & their application) in pharmaceutical industry.
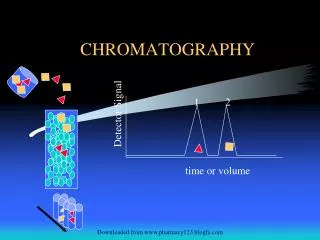
DEFINITION CHROMATOGRAPHY The separation of a mixture by distribution of its components between a mobile and stationary phase over time • mobile phase = solvent • stationary phase = column packing material
HISTORY • Chromatography (from Greek :chromatos -- color , "graphein" -- to write) • 1903 Tswett - plant pigments separated on chalk columns • 1931 Lederer & Kuhn - LC of carotenoids • 1938 TLC and ion exchange • 1950 Reverse phase LC • 1954 Martin & Synge (Nobel Prize) • 1959 Gel permeation • 1965 instrumental LC (Waters)
Purpose of Chromatography • Analytical - determine chemical composition of a sample • Preparative - purify and collect one or more components of a sample
Uses for Chromatography • Real-life examples of uses for chromatography: • Pharmaceutical Company – determine amount of each chemical found in new product • Hospital – detect blood or alcohol levels in a patient’s blood stream • Law Enforcement – to compare a sample found at a crime scene to samples from suspects • Environmental Agency –determine the level of pollutants in the water supply • Manufacturing Plant – to purify a chemical needed to make a product
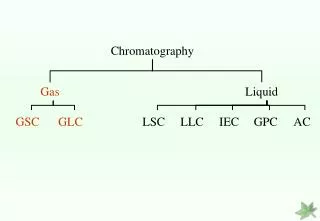
Classification of Methods There are two classification schemes: • mobile phase • attractive forces
Mobile Phase • Gas (GC) • Water (LC) • Organic solvent (LC) • Supercritical fluid (SCFC)
Classification based on Mobile Phase Gas Chromatography Gas - solid Gas - liquid Stationary Phase
Classification based on Mobile Phase Liquid chromatography (LC) Column (gravity flow) Thin layer(adsorption) High performance (pressure flow)
Classification based on Attractive Forces • Adsorption • Ion Exchange • Partition • Size Exclusion
Adsorption Chromatography • Separation based on their adsorption onto the surface of solid (stationary phase). • Normal phase-like separation – Nonpolar mobile phase • for polar non-ionic compounds • Ex; Column chromatography (CC) (K.Turus), TLC, HPLC
Partition Chromatography • solute are separated based on their partition between a liquid mobile phase and a liquid stationary phase coated on a solid support. • Normal – analyte is nonpolar organic; stationary phase MORE polar than the mobile phase • Reverse – analyte is polar organic; stationary phase LESS polar than the mobile phase • Ex : TLC, Paper Chromatography Phase 2 Phase 2 Phase 1 Phase 1
Ion Exchange Chromatography • Use ionic stationary phase • ions separated on the basis of their tendency to displace counter ions adsorbed on stationary phase (Depends on charge, hydration, “solubility”…) • Anionic stationary phases: used for cation separation • Cationic stationary phases : for anion separation • for ionic compounds • - Ex : CC (K.turus), HPLC
Size Exclusion Chromatography • Separation is a result of “trapping” of molecules in the pores of the packing material • Very large molecules can’t get into the pores – unretained • Very small molecules get hung up in to pores for a long time - most retained – longest retention time • stationary phase is a porous matrix • Ex: CC, HPLC
Types of Chromatography • Liquid Chromatography – separates liquid samples with a liquid solvent (mobile phase) and a column composed of solid beads (stationary phase) • Gas Chromatography – separates vaporized samples with a carrier gas (mobile phase) and a column composed of a liquid or of solid beads (stationary phase) • Paper Chromatography – separates dried liquid samples with a liquid solvent (mobile phase) and a paper strip (stationary phase) • Thin-Layer Chromatography – separates dried liquid samples with a liquid solvent (mobile phase) and a glass plate covered with a thin layer of alumina or silica gel (stationary phase)
DEFINITION A chromatographic analytical separation technique for complex mixtures involving the progressive adsorption of the dissolved component onto a special grade of paper.
The certain solvent are used to separate a mixture ex: water, alcohol. • With capillary action the solvent will move up to filter paper. • Movement of a solvent will bring together component that are separated from the mixture. • Every component that are separated will move to several velocity PRINCIPLE
The moving components are depend on :a. Solubility solute in solventb. Intermolecule forcesc. Pore size of filter paperd. Size of soluteAt the end of process, components that are separated will emerge to different distance on filter paper.Rf values are used to identification of each the component.
The retention factor, or Rf, is defined as the distance traveled by the compound divided by the distance traveled by the solvent For example, if a compound travels 2.1 cm and the solvent front travels 2.8 cm, the Rf is 0.75:
Materials List • Beakers or jars • Covers or lids • Solvent (Distilled H2O, Isopropanol) • Graduated cylinder • Filter paper • Sampel (Different colors of pens, plant extract) • Pencil • Ruler • Scissors • Tape
Preparing the solvent solution • Prepare the solvent solution in various concentration: • - 0%, 5%, 10%, 20%, 50%, and 100%
Preparing the Chromatography Strips • Cut filter paper • Draw a line 1 cm above the bottom edge of the strip with the pencil • Label each strip with its corresponding solution • Place a spot from each pen on your starting line
Developing the Chromatograms • Place the strips in the beakers • Make sure the solution does not come above your start line • Keep the beakers covered • Let strips develop until the ascending solution front is about 2 cm from the top of the strip • Remove the strips and let them dry
Spot Detection • Color spot observed by naked eye • Non – color spot color reagent will give specific colors for different compound. Example : • Ninhydrin – a.amino • Iodin dalam etanol –bes (termasuk alkaloid) • AgNO3 berammonia - Karbohidrat
USES OF PAPER CHROMATOGRAPHY • Separation of ink dyes - To compare ink dyes use in any company. 2. Food coloring - To differentiate coloring agent used in food product such as : M&M, Smarties dan Reese candies. 3. Botanist/herbalist - To isolate plant pigment from root and leaves