Polysaccharides
Polysaccharides. www.themegallery.com. By Dr. Batoul Izzularab. 3. 4. Polysaccharides. Polysaccharides are long carbohydrate molecules of monosaccharide units joined together by glycosidic bonds. They range in structure from linear to highly branched.


Polysaccharides
E N D
Presentation Transcript
Polysaccharides www.themegallery.com
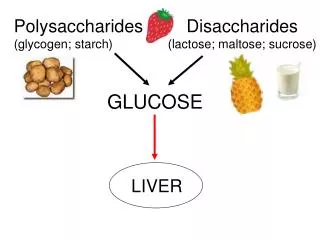
Polysaccharides • Polysaccharides are long carbohydrate molecules of monosaccharide units joined together by glycosidic bonds. They range in structure from linear to highly branched. • When all the monosaccharides in a polysaccharide are the same type, the polysaccharide is called a homopolysaccharide or homoglycan, but when more than one type of monosaccharide is present they are called heteropolysaccharides or heteroglycans.
General properties of polysaccharides • They are formed from great number of monosaccharide units (more than ten unit) attached together by glucosidic bond. • They are high molecular weight compound • All give positive molish test • All are nonreducing sugar • All are optically active compound • All are negative for ozazone test
General properties of polysaccharides • All formed colloid solution i.e. insoluble in water , on hydrolysis they give a great number of monosaccharide unit • They form specific color with iodine e.g. starch gives blue color with iodine • Beside they are important nutritive mitral , they are important in manufacture of • Textile • Artificial silk • Paper
Homopolysaccharide • Starch • It is a homopolymer of glucose forming an α- glucosidic chain, called glucosan or glucan • It is the most important dietary carbohydrate in cereals, potatoes, legumes and other vegetables. • Starch never present in animals tissue
Starch • Starch granules is formed of amylose (13-20%) which has nonbranching helical structure , and amylopectin (80-85%) , which consists of branched chains composed of 24-30 glucose residues united by α1→4 linkages in the chain and by α1→6 linkages at the branched points
Starch • Amylose (13-20%) • The amylose represents the inner part of starch granules. • It has a non-branching helical coil structure, containing only α1→4 glycosidic link. • It gives deep blue color with iodine.
Starch • Amylose • Amylose α1→4 linkage
Starch • Amylopectin (80-85%) • This represents the outer layer of starch granules. • It is a branched chain structure, containing both 1→4glycosidic link (Straight chain and 1-6bond (branch point) • Each chain of amylopectin in composed of 25-30 glucose units linked together by 1-6 bond at the branch points. • Amylopectin gives red color with iodine
Starch • Amylopectin • Amylopectin α1→6 linkages
Starch • Acid hydrolysis of starch produces great number of glucose units • Enzymatic hydrolysis of starch by salivary amylase • Starch →amylodextrin →achrodextrin →maltose
Dextrin • Dextrins are produced during enzymatic hydrolysis of starch by salivary amylase and pancreatic amylase. • They are -like starch- formed of glucose units attached together by 1-4 and 1-6 glycosidic bonds. • Their structure is simpler than starch.
Glycogen • Glycogen is the store of carbohydrates in animal life called "Animal starch". • It is present mainly in the liver and muscles and gives red color with iodine. • Chemically, it a highly branched chain homopolysaccharide.
Glycogen • Each chain is composed of 12-14 glucose units linked by 1-4glycosidic link in the straight chain and 1-6 link in the branch points. It resembles the amylopectin part of starch. • Glycogen store in liver which function as a source of blood glucose maintenance during starvation and in between meals
Cellulose • It is a long straight non-branching chains of glucose units) -D-glucose), attached by1-4 glycosidic link. • The straight chains are cross – linked by hydrogen bonds. • It is the chief constituent of the frame work of plants e.g. leaved vegetables, cotton, wood…etc
Cellulose • Properties: • Non-reducing homopolysaccharide. • Gives no color with iodine. • Not digested in the human GIT, because of the absence of hydrolytic enzymes that attack -link.
Cellulose • Its presence in the diet is important, because it cannot be digested, so it forms the main bulk of stool, stimulating the intestinal movements, and preventing constipation. • Cellulose is digested in the GIT of herbivores, thus acting as source of energy. The GIT of these animals contains microorganisms that produce cellulase enzyme
Inulin • Inulin is a fructosan, formed of -fructose units attached by 1-2 glycosidic link. • It is present in plants e.g. root of dahlias, artichokes and other plants. • It is not digested or metabolized by human GIT. • Inulin clearance is one of the important physiological diagnostic tests for investigation and measurement of glomerular filtration rate (GFR) of the kidney.
Chitin Chitin forms of the shells of crustaceans and exoskeleton of insects. • It is formed of repeated N-acetylglucosamine units
Heteropolysaccharides • Heteropolysaccharides are polysaccharides on hydrolysis give rise into different type of monosaccharide unit i.e. different of monosaccharide units • Agar Agar • Derived from certain sea weeds. It is used as culture media for bacterial growth. The main sugar unit is galactose.
Pectins • These are Heteropolysaccharides containing arabinose, galactose, and galacturonic acid. • They are present in fruits, like orange, lemons, apricots…etc. • Pectins have gellation properties, hence used for treatment of infantile diarrhea.
Glycosaminoglycans (GAG), (mucopolysaccharides( Glycosaminoglycans are Heteropolysaccharides which are present abundantly in the human body.
Glycosaminoglycans (GAG), (mucopolysaccharides( • They are characterized by: • Chemically, they are formed mostly of repeated disaccharides units that contain • amino sugar (N-acetyl glucosamine or N-acetyl galactosamine) • uronic acid (glucuronic acid or its 5 epimer iduronic acid
Glycosaminoglycans (GAG), (mucopolysaccharides( • Most are present extracellulary except heparin. • mostly they act as structural components of the connective tissue e.g. collagen, bones, elastin, …etc • They can also act as lubricants, because they have the property of holding water e.g. synovial fluid and vitreous humor of the eye.
Glycosaminoglycans (GAG), (mucopolysaccharides( • Types: • GAGs are either sulfate free (Hyaluronic acid) or sulfate containing as chondroitin sulfate, heparin, heparan sulfate, keratan sulfate and dermatan sulfate.
Hyaluronic Acid: • Structure: • Repeated disaccharide units, composed of glucuronic acid and N-acetyl glucosamine.
Hyaluronic Acid: • Site • Present in • Synovial fluid. • Vitreous humor of the eye. • Skin and loose connective tissue. • Cartilage • Embyronic tissue.
Hyaluronic Acid: Functions: • Gel made of hyaluronic acid has good resistance to compression, thus it acts as lubricant and shock absorber in the synovial fluid. • It makes extra cellular matrix loose, because of its ability to attract water. • It forms an important part of cementing ground substance.
Hyaluronidase Enzyme • This enzyme hydrolyses hyaluronic acid, thus allowing invasive agents to penetrate the tissue. • It is secreted by certain bacteria to help their invasion to tissue. • Secreted by Cercaria (infective stage of bilharziasis) to help penteration of skin.
Chondroitin Sulfate: • Structure: • Repeated disaccharide units composed of Glucuronic acid and N-acetyl galactosamine sulfate. The sulfate group is either attached to carbon 4 forming chondroitin -4- sulfate or carbon 6 forming chondroitin -6- sulfate.
Chondroitin Sulfate • Site : • Cartilage, tendons, ligaments, bones. • Aorta, skin, cornea. • Functions: • It has a role in binding collagen of cartilage and holding its fibers together. Compressibility of cartilage in weight bearing is due to chondroitin sulfate.
Heparin • Structure: • Disaccharide repeated units of • Iduronic acid with sulfate on C2 • Glucosamine with sulfate on C3 and C6.
Heparin • Site: • In the mast cells (intracellular compound ( • Function: • Anticoagulant • Stimulates the release of lipoprotein lipase enzyme that hydrolyses the absorbed fats.
Heparan Sulfate: • It is like heparin in structure, but it • (1) contains fewer sulfate groups and • (2) Some glucosamines are acetylated. • Function: • It is an extra cellular compounds, entering in the structure of receptors on cell surface. • It participates in cell adhesion and cell-cell interaction.
Keratan Sulfate Structure: • The disaccharide unit is composed of • (1) Galactose with sulfate on C6 and • (2 (N-acetyl glucosamine with sulfate on C6. No uronic acid Site:Cornea and cartilage .It plays a role in corneal transparency.
Dermatan Sulfate Structure: • Disacchride units composed of • L-Iduronic acid • N-acetyl galactosamine with sulfate on C6. • It is widely distributed in animal tissue, resembling chondroitin sulfate and Heparan sulfate. • It has anti thrombotic properties similar to heparin.
Proteoglycans and Glycoproteins: • Both are proteins containing carbohydrates but they contain different sugars, have different structures perform different functions and present in different sites.
Proteoglycans • When proteins are attached to glycosaminoglycans (mucopolysaccharides), the compound is called Proteoglycans. • They serve as ground substance associated with structural elements of tissue e.g. bones, cartilage, elastin, collogen …etc. • The glycosaminoglycans are present as long unbranched chains around a protein core.
These occur in many different situations in fluids and tissues, including cell membranes. They have protein core to which are attached carbohydrate chains which are short, composed of 2-15 sugar units, or often oligosaccharide units, and branched. Glycoproteins (Mucoproteins):
Sugar present in glycoproteins include: Hexoses: Galactose and mannose. Pentoses: Arabinose and xylose. Melthyl pentose: (L-fucose Sialic acid. Acetyl hexosamines, N-acetyl glucosamines and N-acetyl galactosamines. They contain no glucose, no uronic acid or sulfate group. Glycoproteins (Mucoproteins
Functions: Components of extra cellular matrix. Act as biological lubricants in mucin of the GIT, respiratory tract and urogenital tract Components of cell membrane as: Blood group antigens, A, B, AB. Cell surface recognition receptors Glycoproteins (Mucoproteins
Functions: Certain hormones are glycoproteins as Thyroid stimulating hormone (TSH) and human chorionic gonadotrophin HCG. Some enzymes are glycoproteins The immunologic components e.g. Immunoglobulins,complement factors, interferon ...etc are glycoproteins Glycoproteins (Mucoproteins
Thank You! www.themegallery.com