Electron Affinity
Electron Affinity. Ryan Hebling, Jackson Miller, & Libby Takimoto. Definition. Affinity - a liking or attraction to something Electron Affinity - the attraction of an atom to an electron to form a negative ion and the energy change associated with it. Summary.

Electron Affinity
E N D
Presentation Transcript
Electron Affinity Ryan Hebling, Jackson Miller, & Libby Takimoto
Definition Affinity - a liking or attraction to something Electron Affinity - the attraction of an atom to an electron to form a negative ion and the energy change associated with it
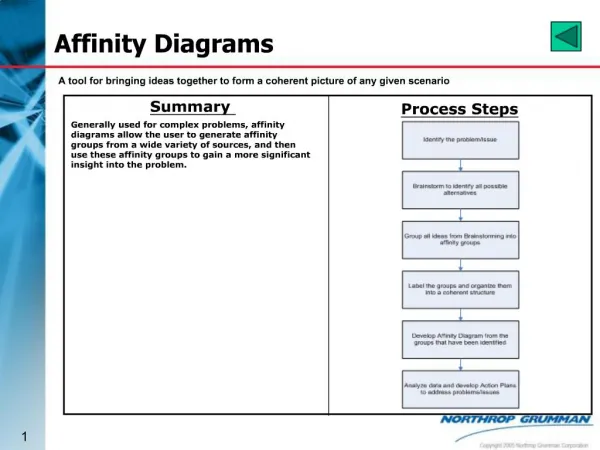
Summary -Electron affinity relates to the formation of negative ions. - X(g) + e- → X-(g) -Change in energy (kJ/mol) of a gaseous neutral mole of atoms when electrons are added. -Corresponds to the possibility of an atom collecting an electron. -Adding an electron to a neutral atom results in a release of energy. -Adding an electron to a negative ion requires more energy than is released, as the electron is being repulsed by the electron already in the atom.
Trends -Present mostly in representative elements -Increases left to right on the table, due to the electrons being closer to the nucleus and having a stronger attraction, thus releasing more energy. -The more valence electrons an atom has, the more likely it will attract an electron to form a stable outer shell. -Although the number of valence electrons increases going down the table, • -Shielding effect when the other electrons in the atom repulse the incoming electron, preventing it from entering the atom. • -Increases bottom to top on the table, because the electrons are positioned closer to the nucleus, and have a greater energy.
Exceptions -Some of the 2nd period elements don’t follow the pattern -Boron, Carbon, Oxygen and Fluorine have lower EA than expected. -While the incoming electron will be close to the nucleus, the small size of the atom concentrates the other electrons in the atom. -This creates a barrier that blocks incoming electrons. -Nitrogen won’t accept another electron because the incoming electron would occupy an orbital that already has an electron in it -This also happens to other elements in group five -Alkaline Earth metals won’t add electrons because they have a filled s subshell -Noble Gases won’t add electrons because they already have a full valence shell
Second Electron Affinity -Group 6 elements have the possibility to gain another electron, while in the gaseous state. -This requires more energy than first electron affinity, due to the atom already having a negative charge. The electron will not enter the atom without assistance.
How it’s Measured - Energy change when an electron is added from the neutral form of an atom, forming a negative ion. - Difficult to measure → only available for a few elements (halogens). - Measure of atom’s tendency to act as an oxidizing agent. - Related to the nature of chemical bonds element forms with.